在脊椎动物中,面部颅骨的形成涉及一系列发育过程,颅骨发育由来自神经嵴、内胚层、中胚层和外胚层的细胞所参与和介导[1]。骨骼成分来自神经嵴,肌肉成分来自体节中胚层。头部同一轴向区域的颅神经嵴和近轴中胚层一起迁移,形成鳃弓或咽弓[2]。脊椎动物胚胎中颅神经嵴、体节中胚层和鳃弓这些组织的高度保守性保障了颅骨发育的稳定性。任何改变这种有序排列的变化都会影响颅面的发育[3]。hox基因对颅面发育具有一定作用,其编码具有同源框的一大类转录因子家族,全称同源异型基因 (homeobox gene),在染色体上成簇排列,其序列中都含有一段保守序列,由180~183个碱基组成,编码60~61个氨基酸,称为同源结构域 (homeodomain,HD)[4,5]。hox基因的表达遵循时间-空间共线性表达模式,其在3'端的基因会最先表达,且表达在躯体的前端[6]。hoxa1a基因是hoxaa基因簇最靠近3'端的基因,是hox基因簇中最先表达的基因,在基因组中发挥着重要的作用。
2005年,Tischfield等[7]报道人类HOXA1基因的纯合缺失会导致阿萨巴斯卡发育不良综合征 (Athabascan brainstem dysgenesis syndrome, ABDS)。其特征表现为面部畸形、面部麻痹、颈动脉形成缺陷等[8,9]。在小鼠中,其直系同源基因Hoxa1是最先在小鼠胚胎中表达的Hox基因[10],Hoxa1是神经嵴细胞的迁移所必需的,Hoxa1的缺失会影响颅神经嵴细胞,从而导致小鼠咽弓缺陷,使颅面发育异常和弓动脉形成缺陷[11]。Hoxa1与Hoxa2共同缺失会导致小鼠产生颅骨缺陷及腭裂[3]。Hoxa1在小鼠和人类的神经嵴细胞迁移、后脑模式、心脏和耳朵发育中都具有一定的功能作用[12]。但是,Hoxa1基因对颅面骨骼发育影响的作用机制尚未研究清楚。hoxa1a在斑马鱼中仅有报道其mRNA在胚胎早期的脑部神经元中有表达,推测其可能影响脑的发育[13]。颅面骨骼的基本信号通路和细胞事件被证明从鱼到人是高度保守的[1]。模式生物斑马鱼具有幼体阶段骨骼简单、生长发育快速、体型小利于染色成像等优势,对于研究面部颅骨发育十分有利。本文为ABDS疾病研究提供一个良好的斑马鱼模型,我们运用CRISPR/Cas9技术在斑马鱼中构建了缺失hoxa1a基因的纯合突变体,对hoxa1a基因功能进行初探。为探究hoxa1a在颅骨发育过程中的作用机制奠定了基础,为ABDS疾病的致病机制研究提供了一些新的思路。
1 材料与方法
1.1 斑马鱼的饲养
按照上海海洋大学动物伦理相关规定 (IACUC SHOU-DW-2021-042)对斑马鱼进行养殖与实验,本实验所用的斑马鱼均为AB品系的。我们拥有完善的斑马鱼养殖设备,其生活的环境是经过紫外线及曝气处理过的循环水系统,水温为28.5℃,pH和电导率均在正常指标内,光照也是严格按照要求每天黑暗10 h、光照14 h。斑马鱼在专门的产卵缸中交配产卵,将收集的斑马鱼卵放于28.5℃的恒温培养箱培养。
1.2 设备仪器及试剂
本实验所用仪器主要包括Warner Instruments公司的显微注射仪、Eppendorf公司的离心机、Bio-Rad公司的PCR仪和Bio-Rad公司的凝胶成像系统等。主要试剂包括Taq酶、体外转录试剂盒、感受态细胞、T7E1酶等均来自Vazyme公司,Cas9蛋白来自GenScript公司,阿尔新蓝、茜素红及常规试剂和引物均来自Sangon Biotech公司。
1.3 质粒来源
实验所用的体外合成 gRNA pUC19-scaffold plasmid 质粒来自于北京大学分子医学研究所熊敬维教授实验室惠赠。
1.4 敲除靶点设计及检测引物
在Ensembl网站 (http://asia.ensembl.org/Danio_rerio/Info/Index) 上下载斑马鱼hoxa1a基因的序列。在ZIFIT网站(http://zifit.partners.org/zifit/)上选取合适的gRNA,序列为5'-GGTGGAGATGGAGGTAGCGG-3';在NCBI网站上进行BLAST确定敲除靶点特异性,并设计检测引物。上游引物F: 5'-TATCACTAGCGCCCGAACAC-3';下游引物R: 5'-TCACAGACGATTCCACGTCC-3'。
1.5 显微注射敲除hoxa1a基因
将合成好的gRNA与Cas9蛋白按照终浓度gRNA 100 ng/μL,Cas9蛋白800 ng/μL混合后,注射到斑马鱼1细胞时期的动物极中,48 h后取3组胚胎,每组5枚进行T7E1酶切效率检测,利用2%的琼脂糖凝胶进行电泳实验,出现三条条带即为酶切成功,将酶切成功的胚胎养起来作为F0。
1.6 筛选获得hoxa1a纯合突变体
待F0长大后与野生型外交产生F1,待F1小鱼长大对其剪尾鉴定其基因型,将其杂合子自交,产生的后代作为F2,F2中符合孟德尔第一遗传定律,将会有1/4纯合突变体,对F2成鱼进行剪尾检测,送公司测序,最终筛选得到缺失了hoxa1a基因的纯合突变体。
1.7 阿尔新蓝软骨染色
收集样品,先用4%多聚甲醛在4℃固定过夜;后用PBST洗涤几次;配制阿尔新蓝染色液,将阿尔新蓝粉末溶于80%乙醇+20%乙酸(V乙醇∶V乙酸=4∶1)的溶剂中,终浓度为0.02%,将样品放入阿尔新蓝溶液中,染色6~24 h,根据样品大小而定;染色结束后使用100%乙醇进行洗涤,然后进行梯度复水,每次30 min;使用漂白液去除色素,用PBST清洗;用0.1%胰蛋白酶消化组织,将组织消化至透明,骨骼清晰可见为止;最后用甘油进行梯度透化,保存在100%甘油中,进行拍照。
1.8 茜素红硬骨染色
按照上述阿尔新蓝染色步骤,将样品消化至透明;将茜素红粉末溶于0.5% KOH溶液中,配制成0.000 2%的茜素红染色液,再将样品放入染色液中,染色至深紫色;移去染色液,依次使用25%甘油、50%甘油、75%甘油、100%甘油进行梯度透化,甘油使用0.25% KOH溶液进行稀释,透化至透明即可,最后保存于100%甘油中,进行拍照。
2 结果与分析
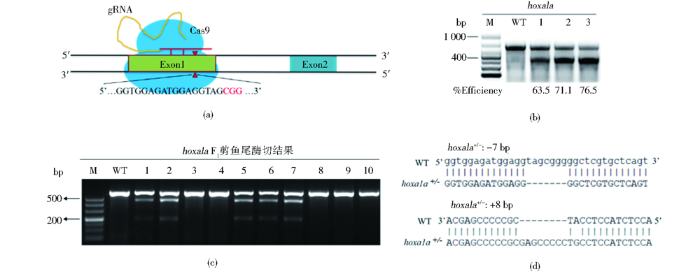
2.1 利用CRISPR/Cas9编辑斑马鱼hoxa1a基因
图1
图1
利用CRISPR/Cas9系统编辑斑马鱼hoxa1a基因
Fig.1
Using CRISPR/Cas9 system to edit zebrafish hoxa1a gene
(a) CRISPR/Cas9 knockout pattern (b) Knockout efficiency of F0 was 70% on average (c) Screening for F1 zebrafish with inherited mutation using T7E1 (d) F1 heterozygous sequence alignment, screening +8 bp heterozygous and -7 bp heterozygous
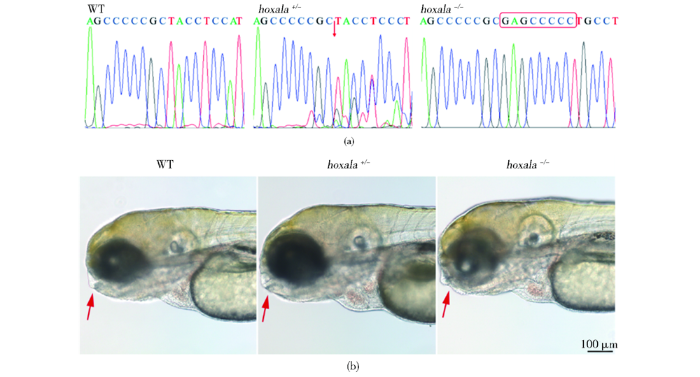
2.2 成功获得hoxa1a基因纯合突变体
图2
图2
成功得到hoxa1a的纯合突变体斑马鱼,并发现面部骨骼发育畸形
Fig.2
A homozygous hoxa1a-/- zebrafish with abnormal facial development was successfully established
(a) Comparison of sequencing peak maps of wildtype, hoxa1a+/- and hoxa1a-/- zebrafish. The red arrows indicate the position at which the mutation starts, and the red boxes indicate the inserted bases (b) Phenotype observations showed that the wildtype and heterozygous mutant had normal faces, while the homozygous mutant faces skeletal were abnormal at 5 dpf. As shown by the red arrow, Scale bar: 100 μm
2.3 hoxa1a突变后的蛋白质结构
hoxa1a全长329个氨基酸,C端的255~284位氨基酸包含一个DNA结合域。敲除后,其插入的8个碱基产生了移码突变,使得hoxa1a的氨基酸序列在17位后发生改变,并提前出现终止密码子终止转录,仅剩N端46个氨基酸。突变后,hoxa1a丧失了大部分氨基酸序列以及保守的DNA结合功能性结构域,无法结合DNA,失去转录调控的作用,因此推测hoxa1a纯合突变体中插入了8个碱基导致hoxa1a基因丧失蛋白质功能(图3)。
图3
图3
hoxa1a突变氨基酸序列及其蛋白质结构
Fig.3
Amino acids sequence and protein structure of hoxa1a mutation
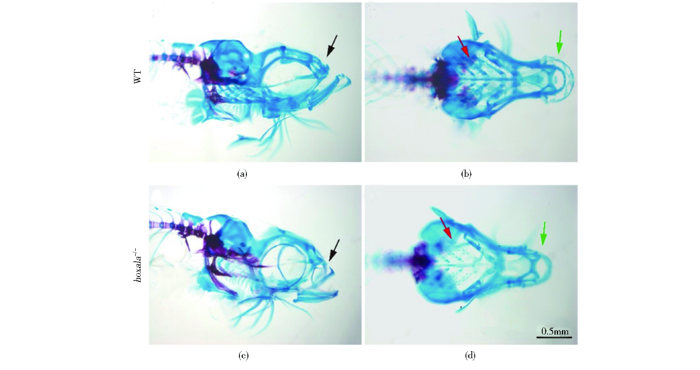
2.4 hoxa1a突变导致成鱼面部颅骨异常且筛骨板断裂
图4
图4
斑马鱼成鱼硬骨染色表明hoxa1a纯合突变体面部颅骨发育缺陷、筛骨板断裂
Fig.4
Alizarin red staining of adult hoxa1a-/- zebrafish showed defective skull development and rupture of ethmoid plate
(a),(b) Alizarin red staining of wildtype at 3 months showed normal ethmoid plate (c),(d) Alizarin red staining of homozygous hoxa1a mutant at 3 months showed rupture of ethmoid plate, as shown by the red arrow (e),(f) Alizarin red staining of wildtype 1 year old wildtype zebrafish showed normal ethmoid plates (g),(h) Alizarin Red Staining of homozygous hoxa1a mutant zebrafish of 1 year old indicated a rupture of the ethmoid plate, as shown by the red arrow. And the hoxa1a-/- had abnormal mouth shape, as shown by the green arrow. a, c, e, g scale bar: 0.5 mm; b, d, f, h scale bar: 1 mm
2.5 hoxa1a突变体幼鱼鳃弓与筛骨板发育畸形
图5
图5
hoxa1a缺失导致斑马鱼幼鱼面部软骨发育缺陷、筛骨板断裂
Fig.5
The loss of hoxa1a resulted in defective facial cartilage development and ethmoid plate rupture in juvenile zebrafish
(a),(b) The results of cartilage staining of wildtype juvenile zebrafish at 1 month showed normal ethmoid plate and normal development of pharyngeal arch (c),(d) The cartilage staining results of hoxa1a-/- at 1 month showed that the ethmoid plate was broken, as shown by the black arrow. The gill arch was lightly stained and was abnormal development, as shown by the red arrow. There was also a loss of cartilage at the anterior end of the first pharyngeal arch, as shown by the green arrow. Scale bar: 0.5 mm
3 讨论
研究表明人类HOXA1的突变,导致阿萨巴斯卡发育不良综合征,患者会出现颅面畸形和面部麻痹、颈动脉形成缺陷等症状;小鼠中Hoxa1的缺失,会影响小鼠颅神经发育,导致头骨骨骼缺陷、颅骨发育缺陷、咽弓缺陷和弓动脉形成缺陷[14]。2001年,Shih 等[13]报道,斑马鱼hoxa1a基因的mRNA在脑部神经元中表达[15],而脑部神经元与颅骨发育有关。本研究成功在斑马鱼中构建了hoxa1a缺失的纯合突变体,突变体出现了面部颅骨发育异常、鳃弓异常等表型,与小鼠和人类中报道的表型相符。本文成功构建了ABDS斑马鱼疾病模型,后续对其hoxa1a功能的深入研究,将有助于阐明hoxa1a的作用机制,有助于ABDS疾病的致病机制的研究与疾病的治疗。
颅骨发育和咽弓发育与hox基因密切相关,在小鼠中,Hoxa1与Hoxb1同时缺失会导致咽弓发育缺陷[11]。Hoxa7基因缺失[16]、Hoxb6基因缺失[17]、Hoxb7基因缺失[18]、Hoxd4基因缺失[19],这些基因的缺失均会引起小鼠颅骨发育异常,出现腭裂表型。斑马鱼基因组在进化过程中经历了3次加倍,导致斑马鱼有7个hox基因簇,48个hox基因[20]。基因簇之间和基因之间可能存在功能冗余和功能分化现象。本文首先利用CRISPR/Cas9技术在斑马鱼中构建了hoxa1a缺失的突变体,研究其对颅骨发育的影响。后续可以利用该技术构建相关基因的缺失突变体,以探讨hox基因在颅骨发育过程中的作用机制。
本研究通过CRISPR/Cas9技术成功构建缺失了hoxa1a基因的斑马鱼突变体,并观察到纯合突变体颅面骨骼发育异常。通过阿尔新蓝软骨染色和茜素红硬骨染色,进一步发现突变体存在筛骨板断裂、鳃弓发育畸形等表型。hoxa1a基因突变体的构建将为ABDS疾病研究提供一个良好的疾病模型,为ABDS致病机制的研究提供新的思路。
参考文献
Zebrafish craniofacial development: a window into early patterning
Neural crest and mesoderm lineage-dependent gene expression in orofacial development
The present study utilizes a combination of genetic labeling/selective isolation of pluripotent embryonic progenitor cells, and oligonucleotide-based microarray technology, to delineate and compare the "molecular fingerprint" of two mesenchymal cell populations from distinct lineages in the developing embryonic orofacial region. The first branchial arches-bi-lateral tissue primordia that flank the primitive oral cavity-are populated by pluripotent mesenchymal cells from two different lineages: neural crest (neuroectoderm)- and mesoderm-derived mesenchymal cells. These cells give rise to all of the connective tissue elements (bone, cartilage, smooth and skeletal muscle, dentin) of the orofacial region (maxillary and mandibular portion), as well as neurons and glia associated with the cranial ganglia, among other tissues. In the present study, neural crest- and mesoderm-derived mesenchymal cells were selectively isolated from the first branchial arch of gestational day 9.5 mouse embryos using laser capture microdissection (LCM). The two different embryonic cell lineages were distinguished through utilization of a novel two component transgenic mouse model (Wnt1Cre/ZEG) in which the neural crest cells and their derivatives are indelibly marked (i.e., expressing enhanced green fluorescent protein, EGFP) throughout the pre- and post-natal lifespan of the organism. EGFP-labeled neural crest-derived, and non-fluorescent mesoderm-derived mesenchymal cells from the first branchial arch were visualized in frozen tissue sections from gestational day 9.5 mouse embryos and independently isolated by LCM under epifluorescence optics. RNA was extracted from the two populations of LCM-procured cells, and amplified by double-stranded cDNA synthesis and in vitro transcription. Gene expression profiles of the two progenitor cell populations were generated via hybridization of the cell-type specific cRNA samples to oligo-based GeneChip microarrays. Comparison of gene expression profiles of neural crest- and mesoderm-derived mesenchymal cells from the first branchial arch revealed over 140 genes that exhibited statistically significant differential levels of expression. The gene products of many of these differentially expressed genes have previously been linked to the development of mesoderm- or neural crest-derived tissues in the embryo. Interestingly, however, hitherto uncharacterized coding sequences with highly significant differences in expression between the two embryonic progenitor cell types were also identified. These lineage-dependent mesenchymal cell molecular fingerprints offer the opportunity to elucidate additional mechanisms governing cellular growth, differentiation, and morphogenesis of the embryonic orofacial region. The chemokine stromal cell-derived factor 1, (SDF-1), was found to exhibit greater expression in mesoderm-derived mesenchyme in the branchial arch when compared with neurectoderm, suggesting a possible chemotactic role for SDF-1 in guiding the migratory neural crest cells to their destination. The novel combination of genetic labeling of the neural crest cell population by EGFP coupled with isolation of cells by LCM for gene expression analysis has enabled, for the first time, the generation of gene expression profiles of distinct embryonic cell lineages.
Compensatory defects associated with mutations in Hoxa1 restore normal palatogenesis to Hoxa2 mutants
Reassessing the role of hox genes during vertebrate development and evolution
The Hox genes and their roles in oncogenesis
The regulation of Hox gene expression during animal development
Homozygous HOXA1 mutations disrupt human brainstem, inner ear, cardiovascular and cognitive development
We identified homozygous truncating mutations in HOXA1 in three genetically isolated human populations. The resulting phenotype includes horizontal gaze abnormalities, deafness, facial weakness, hypoventilation, vascular malformations of the internal carotid arteries and cardiac outflow tract, mental retardation and autism spectrum disorder. This is the first report to our knowledge of viable homozygous truncating mutations in any human HOX gene and of a mendelian disorder resulting from mutations in a human HOX gene critical for development of the central nervous system.
Bilateral complete labyrinthine aplasia with bilateral internal carotid artery aplasia, developmental delay, and gaze abnormalities: a presumptive case of a rare HOXA1 mutation syndrome
Athabascan brainstem dysgenesis syndrome
A distinct Hox code for the branchial region of the vertebrate head
Hoxa1 and Hoxb1 are required for pharyngeal arch artery development
Hoxa1 targets signaling pathways during neural differentiation of ES cells and mouse embryogenesis
Hoxa1 has important functional roles in neural crest specification, hindbrain patterning and heart and ear development, yet the enhancers and genes that are targeted by Hoxa1 are largely unknown. In this study, we performed a comprehensive analysis of Hoxa1 target genes using genome-wide Hoxa1 binding data in mouse ES cells differentiated with retinoic acid (RA) into neural fates in combination with differential gene expression analysis in Hoxa1 gain- and loss-of-function mouse and zebrafish embryos. Our analyses reveal that Hoxa1-bound regions show epigenetic marks of enhancers, occupancy of Hox cofactors and differential expression of nearby genes, suggesting that these regions are enriched for enhancers. In support of this, 80 of them mapped to regions with known reporter activity in transgenic mouse embryos based on the Vista enhancer database. Two additional enhancers in Dok5 and Wls1 were shown to mediate neural expression in developing mouse and zebrafish. Overall, our analysis of the putative target genes indicate that Hoxa1 has input to components of major signaling pathways, including Wnt, TGF-β, Hedgehog and Hippo, and frequently does so by targeting multiple components of a pathway such as secreted inhibitors, ligands, receptors and down-stream components. We also identified genes implicated in heart and ear development, neural crest migration and neuronal patterning and differentiation, which may underlie major Hoxa1 mutant phenotypes. Finally, we found evidence for a high degree of evolutionary conservation of many binding regions and downstream targets of Hoxa1 between mouse and zebrafish. Our genome-wide analyses in ES cells suggests that we have enriched for in vivo relevant target genes and pathways associated with functional roles of Hoxa1 in mouse development.Copyright © 2017 Elsevier Inc. All rights reserved.
Expression of zebrafish Hoxa1a in neuronal cells of the midbrain and anterior hindbrain
The expression pattern of zebrafish hoxa1a mRNA during embryonic development was studied. Herein, we show that hoxa1a mRNA is expressed in the ventral region of both the midbrain and anterior hindbrain during the developmental period from the pharyngula to the protruding-mouth stages via whole-mount in situ hybridization. Furthermore, double-labeling with anti-zHu antibody confirms that the zebrafish hoxa1a gene is expressed in neuronal cells. The observed temporal and spatial distributions of zebrafish hoxa1a mRNA differ greatly from the expression patterns of zebrafish hoxb1a and hoxb1b paralagous genes. In addition, in embryos injected with mouse ihh mRNA, hoxa1a-expressing cells increase in number with a dorsalized expression pattern in the midbrain.
Disruption of the Hox-1.6 homeobox gene results in defects in a region corresponding to its rostral domain of expression
The Hox-1.6 gene disrupted in embryonic stem cells by homologous recombination was introduced into the mouse germline. Heterozygous mice were normal, but homozygous mice died at birth from anoxia and had numerous defects that were centered at the level of rhombomeres 4 to 7 and included delayed hindbrain neural tube closure, absence of certain cranial nerves and ganglia, and malformed inner ears and bones of the skull. Thus, Hox-1.6 is involved in regional specification along the rostrocaudal axis, but only in its most rostral domain of expression. Hox-1.6 appears to specify neurogenic neural crest cells prior to specification of mesenchymal neural crest cells by Hox-1.5. Thus, within the same region of the presumptive hindbrain, two HOX-1 genes are involved in the patterning of two different populations of neural crest cells. The implication of these results for the function of the Hox network during mouse embryogenesis is discussed.
Conserved expression of Hoxa1 in neurons at the ventral forebrain/midbrain boundary of vertebrates
The previously described expression patterns of zebrafish and mouse Hoxa1 genes are seemingly very disparate, with mouse Hoxa1 expressed in the gastrula stage hindbrain and the orthologous zebrafish hoxa1a gene expressed in cell clusters within the ventral forebrain and midbrain. To investigate the evolution of Hox gene deployment within the vertebrate CNS, we have performed a comparative expression analysis of Hoxa1 orthologs in a range of vertebrate species, comprising representatives from the two major lineages of vertebrates (actinopterygians and sarcopterygians). We find that fore/midbrain expression of hoxa1a is conserved within the teleosts, as it is shared by the ostariophysan teleost zebrafish (Danio rerio) and the distantly related acanthopterygian teleost medaka (Oryzias latipes). Furthermore, we find that in addition to the described gastrula stage hindbrain expression of mouse Hoxa1, there is a previously unreported neurula stage expression domain, again located more anteriorly at the ventral fore/midbrain boundary. A two-phase expression profile in early hindbrain and later fore/midbrain is shared by the other tetrapod model organisms chick and Xenopus. We show that the anterior Hoxa1 expression domain is localized to the anterior terminus of the medial longitudinal fasciculus (MLF) in mouse, chick, and zebrafish. These findings suggest that anterior expression of Hoxa1 is a primitive characteristic that is shared by the two major vertebrate lineages. We conclude that Hox gene expression within the vertebrate CNS is not confined exclusively to the segmented hindbrain and spinal cord, but rather that a presumptive fore/midbrain expression domain arose early in vertebrate origins and has been conserved for at least 400 million years.
Craniofacial abnormalities induced by ectopic expression of the homeobox gene Hox-1.1 in transgenic mice
Hox-1.1 is a murine homeobox-containing gene expressed in a time- and cell-specific manner during embryogenesis. We have generated transgenic mice that ectopically express Hox-1.1 from the chicken beta-actin promoter. In these mice Hox-1.1 expression was changed to an almost ubiquitous pattern. Ectopic expression of Hox-1.1 leads to death of the transgenic animals shortly after birth and is associated with multiple craniofacial anomalies, such as cleft palate, open eyes at birth, and nonfused pinnae. This phenotype is similar to the effects seen after systemic administration of retinoic acid during gestation. This suggests that retinoic acid embryopathy and the specific developmental defects caused by ectopic expression of a potential developmental control gene share a common pathogenic mechanism.
Dominant mutation of the murine Hox-2.2 gene results in developmental abnormalities
Ectopic expression of Hox-2.3 induces craniofacial and skeletal malformations in transgenic mice
To better understand the role of the Hox-2.3 murine homeobox gene during development, a dominant gain-of-function mutation was generated. The developmental malformations that resulted when the chicken beta-actin promoter was used to direct widespread expression of the Hox-2.3 gene in transgenic mice included early postnatal death as well as craniofacial abnormalities, including open eyes and cleft palate. Ventricular septal defects were also observed in the hearts of three transgenic mice. Skeletal malformations were seen in the bones of the craniocervical transition, with the occipital, basisphenoid, and atlas bones deficient or misshapen. Interestingly, one mutant exhibited an extra pair of ribs as well as alterations in cervical vertebrae identities. Some of the malformations observed in Hox-2.3 gain-of-function mutants overlap with those seen in Hox-1.1 and Hox-2.2 misexpression mutants which suggests functional similarities between paralogous homeobox genes. The results of these experiments are consistent with a role for Hox-2.3 in specifying positional information during development.
Homeotic transformation of the occipital bones of the skull by ectopic expression of a homeobox gene