“生物3D打印”是一项在计算机辅助设计下的增材制造技术[3],它以加工包括生物材料、生长因子和细胞等的活性材料为主要内容,以修复和重建具有复杂功能的人体组织和器官为目的,所以它也是跨学科、跨领域的新型再生医学工程技术。以技术产生时间和材料生物学性能为依据,生物 3D 打印技术发展可分为四个阶段[4]:打印材料无生物相容性要求,如体外医疗器械[5];打印材料有生物相容性要求,但不可降解,如金属和陶瓷等永久性植入物;打印材料有生物相容性要求,且能被降解,如可促进组织再生的植入物;使用活细胞、蛋白及其他细胞外基质作为材料,打印三维生物结构体,甚至包括最新发展出的、增加时间维度的可打印自我组装材料的 4D 打印。细胞打印技术是生物3D打印发展的第四阶段,它是利用医学成像技术以及 3D 建模软件建立数字模型,将细胞和生物材料依据该数字模型完成精确堆积,并最终形成形状复杂的 3D 细胞结构[6,7]。细胞打印技术主要有激光引导直写、立体光刻印刷、生物绘图/生物三维打印、直接三维受控组装、喷墨打印(也可被称作微滴法)、批量细胞打印等技术[8,9]。本文针对基于喷墨打印原理的压电式微喷技术,分析其对细胞打印的影响、实施方式以及实际应用。
1 压电式微喷技术
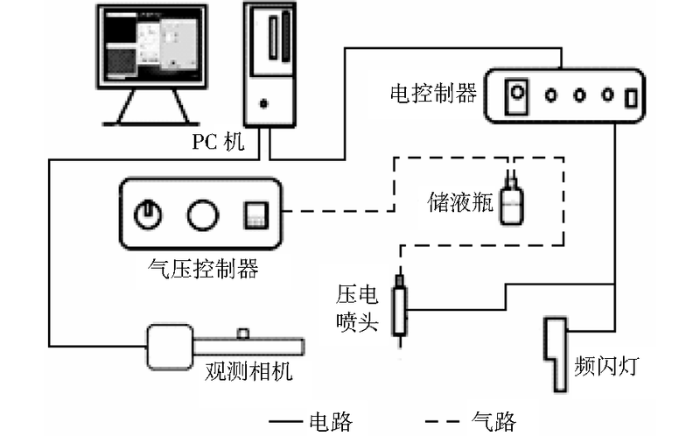
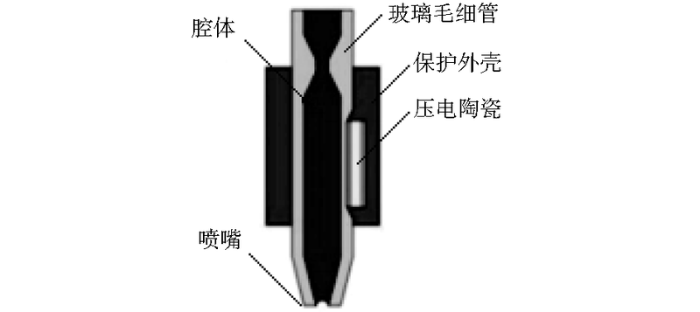
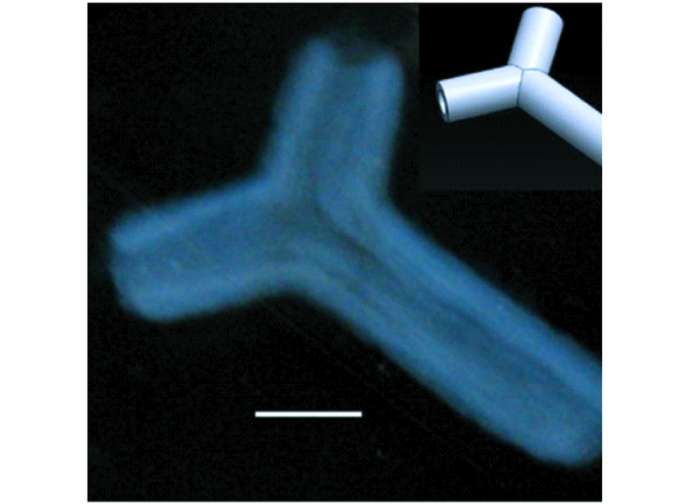
微喷射是微流体控制应用的一个重要体现,压电式微喷打印的原理是利用压电陶瓷材料的伸缩形变行为使喷嘴中“墨汁”喷射出去而形成液滴[9,10]。压电式微喷打印系统如图1所示,主要由电控制器、气控制器、压电喷头和视觉观测组件构成。其中压电喷头如图2所示,喷头材料为玻璃毛细管,其中部外侧有压电陶瓷材料PZT(piezoelectric transducer)。压电式微喷打印系统喷射机理为:气压控制器产生的负压将喷射油墨平衡于喷嘴位置;电驱动控制器施加脉冲波形,脉冲电压上升和保续过程中,喷头内部的压电陶瓷材料产生微弱的形变,形变造成压电陶瓷材料接触附近的玻璃毛细管壁并形成一种“声波”[11],使得喷嘴处的溶液被挤压而喷射出去;当电压下降时,压电陶瓷因形变减缓而放松,玻璃毛细管膨胀,喷嘴处的墨水凹陷而“剪断”挤出的溶液,在墨水表面张力作用下,挤出的溶液将会逐渐聚集形成单一液滴,从而实现满足一定规律的喷射打印。
图1
图1
压电式微喷打印系统原理图
Fig.1
Principle diagram of piezoelectric micro-jet printing system
图2
微喷技术最早应用于微纳电子印刷、生物传感器等工程制造领域,随着技术的不断深入,近年来微喷技术在获取生物细胞和打印器官组织等生物领域的应用取得了快速发展。蔡仁烨[12]利用自主研制的细胞打印装置,基于压电喷射成功打印酵母细胞,并分析了脉冲电压、生物油墨浓度、粘度和缓冲介质等对生物细胞打印成活率的影响; Xu C等[13]研究了成纤维细胞和海藻酸钠混合的生物油墨的液滴生成过程;Kim Y K等[14]使用压电喷头成功获取老鼠成纤维细胞和人胚肾细胞,并通过后期培养、增殖验证了压电微喷技术对哺乳动物细胞打印成活不会造成损伤的观点;Kim J D等[15]用压电式微喷射技术打印人类脂肪干细胞;Ng W L等[16]研究并验证了新生儿包皮成纤维细胞HFF(human foreskin fibroblast)的打印可行性。在组织器官打印领域,Lee J H等[17]报道了一种微流体三维骨骼模型,在该模型制备过程中,微喷技术制备了含有抗生素和双相磷酸钙纳米颗粒的微结构,并与微流芯片和细胞培养相结合,加速了骨骼和植入物的结合同时可以防止细菌感染;Zhang J等[18]通过自主研制的微喷设备在载玻片上精确制备了海藻酸钠微型结构,与相应的细胞阵列结构结合形成微流体芯片,用以模拟体内环境。
可见,压电微喷技术在生物细胞打印领域已取得了一系列进展,为细胞获取、组织器官再生等研究和应用提供了基础。
2 压电式微喷技术对细胞打印的影响
压电式微喷实施细胞打印的影响因素包括微喷技术条件和生物油墨性质。
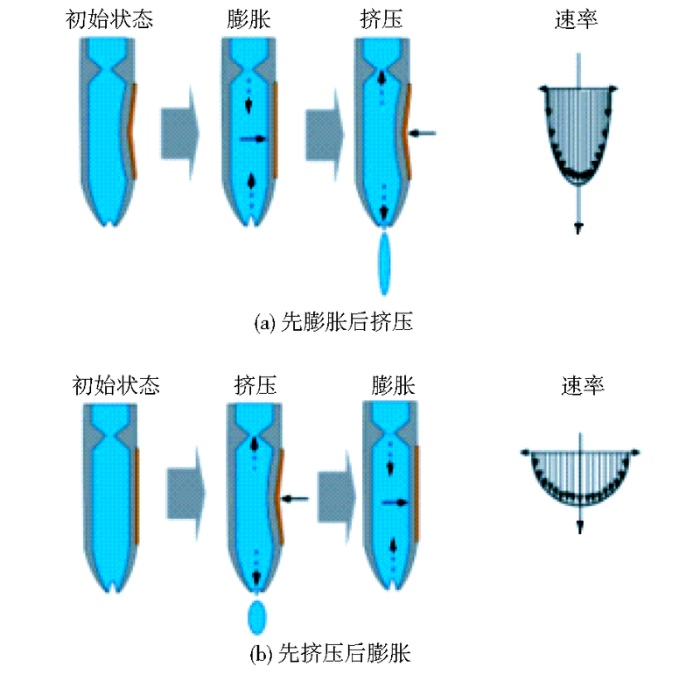
2.1 压电驱动方式对细胞打印的影响
图3
Yamaguchi S等[20]对昆虫细胞sf9(spodoptera frugiperda cell)进行打印实验分析,证明“先挤压后膨胀”的方式更易于产生单细胞液滴(100个液滴中全部只含有一个细胞),且与未进行喷射的细胞对比,发现细胞存活率都在90%左右,喷射过程对细胞存活率不会造成明显影响。可见,对于细胞打印尤其是单细胞打印而言,“先挤压后膨胀”的压电驱动方式更合适。
2.2 压电参数对细胞打印的影响
打印过程中图文信息的传输与喷头中压电陶瓷激励脉冲的匹配情况影响微喷射的稳定性和准确性[21]。如前所述,压电喷头的驱动力来自于压电陶瓷材料的形变量,而压电陶瓷的形变量主要是由驱动电源的脉冲电压所决定的,所以,压电喷头的工作性能直接取决于电控制器提供的脉冲电压及频率,这直接影响到喷头最终形成液滴的尺寸、喷射速度、液滴均匀性和液滴脱离喷嘴后的直线性等,最终影响细胞打印效果。Zhang M等[22]利用3T3鼠成纤维细胞和海藻酸钠混合溶液研究了脉冲电压对细胞分布的影响,结果表明:脉冲电压为40V时,细胞实际分布值与理论分布值相差3%;脉冲电压为50V时,则相差18%。另外, Saunders R E等[23]利用成纤维细胞溶液研究了脉冲电压与电压持续时间对细胞活性的影响,得出的结论是:脉冲电压和电压持续时间变大,液滴速度增高,夹裹细胞的液滴与基板产生的撞击力增大,降低细胞存活率。可见,压电参数对细胞打印有着直接影响。
2.3 脉冲驱动电压波形对细胞打印的影响
电控制器为压电喷头提供的驱动电压波形有多种形态,Gan H Y等[24]研究了单极波、双极波、M形波、W形波等不同波形驱动电压对液滴大小的影响。结果表明:双极波更加适合于牛顿流体或近似牛顿流体,对液滴体积减小影响更大;W形波和M形波可调节范围小,但对非牛顿流体影响十分明显,能够减小液滴体积,从而降低线宽。压电微喷最常采用的波形为图4所示的双极梯形波:X轴表示时间(μs),Y轴表示脉冲电压值(V),参数包括脉冲电压幅值Dwell Voltage(V)和Echo Voltage(V)、正负电压保持时间Dwell Time(μs)和Echo Time(μs)、脉冲电压上升时间Rise Time(μs)、脉冲电压下降时间Fall Time(μs)。
图4
随着微喷技术在细胞打印中的应用,为了满足越来越高的精度要求和打印很小的细胞液滴,可以通过使用较小直径喷嘴实现。但喷嘴的直径过小会导致喷嘴容易堵塞,大大降低打印过程的可靠性和重复性。为此,Kwon K S等[25]基于波传导理论提出了一种微喷打印的高速波形设计方法,经过比较发现,对于高速微喷射打印而言,改进脉冲波形可以有效地抑制喷头喷射后的残余压力波,提高打印的稳定性。Lee Y I等[26]通过压电微喷打印在聚酰亚胺表面形成精细线条图案,为了提高打印后的分辨率,通过改进脉冲波形的形态设计来缩短喷头内压电陶瓷的形变周期,进而辅助喷射出速度更快、精度更高的液滴。可见,对于细胞打印而言,改进脉冲驱动电压波形,可以通过改变压电陶瓷的形变频率,改善因细胞油墨黏度较高而出现堵塞不喷的现象,同时在一定程度上提高细胞打印的速度[27]。
2.4 生物细胞油墨对细胞打印的影响
生物细胞油墨对打印液滴稳定性影响研究已有一定成果。Fromm J E [28]对不同溶液进行测试分析,针对打印油墨的物理性质引入了重要无量纲参数Z(与溶液密度、表面张力、粘度和喷嘴直径有关),并预测Z>2时方能形成液滴进行喷射。Reis N等[29]和Derby B [30]对此进行了证明,发现油墨Z值在1~10之间可以形成稳定液滴、进行喷墨打印。Jang D等[31]在此基础上,通过检测稳定液滴形成过程中动力学的变化,在分析打印特性(如:单自由度、最小距离、位置精度、最大喷射频率)后,对Z值进行了重新精确定义:4≤Z≤14,即当溶液Z值低于4时,会出现拖尾情况难以使挤出的溶液聚集成稳定液滴;当Z值大于14时则不能形成稳定液滴进行打印。Chahal D等[32]则具体利用成纤维细胞与海藻酸钠和聚苯乙烯混合溶液研究了浓度对液滴稳定性影响:随着生物细胞油墨浓度增大,液滴的体积和喷射速度增加,能抑制卫星液滴的生成,而液滴拖尾消除时间增大,但与颗粒悬浮溶液相比,生物细胞油墨喷射形成的液滴体积和喷射速度相对较小。Moon S等[33]、Liberski A R等[34]、Cheng E等[35,36]以乳腺癌细胞MCF-7与Ficoll PM400和PBS(phosphate buffer saline,磷酸盐缓冲液)混合溶液为材料研究了细胞油墨特性对细胞分布的影响:微喷射能精确控制液滴大小,但液滴中细胞分布并不均匀,不符合泊松分布;改变细胞油墨的粘度可以改善细胞沉积;细胞在压电喷嘴处沉积会对最终液滴中细胞分布造成影响,且容易堵塞喷嘴;在细胞油墨中加入悬浮液可改变细胞油墨流变特性,使细胞均匀分散、细胞分布近似于泊松分布,降低细胞聚集、堵塞喷嘴的现象。表1列出了几种生物油墨对细胞打印的影响。
表1 几种生物油墨对细胞打印的影响
Table 1
| 生物油墨 | 打印细胞 | 研究目的 | 实验结果 | 参考文献 |
|---|---|---|---|---|
| 聚乙烯吡咯烷 | 成纤维细胞 | 聚合物浓度对Z值和细胞打印稳定性的影响。 | 通过改变聚合物浓度调节Z值可以提高细胞打印稳定性和细胞存活率。 | [16] |
| 海藻酸钠和聚苯乙烯混合液 | 成纤维细胞 | 生物油墨浓度对打印细胞存活率的影响。 | 生物油墨浓度降低,打印细胞存活率相应提高。 | [32] |
| 聚蔗糖和磷酸盐缓冲溶液 | 乳腺癌细胞 | 生物油墨流变特性对细胞打印过程的影响。 | 改善生物油墨流变性可以降低细胞聚集、沉积现象。 | [33-36] |
| 无内毒素低酰基结冷胶悬浮液 | 小鼠成肌细胞 | 生物油墨流变特性对打印细胞存活率的影响。 | 生物油墨流变性好,则打印的细胞存活率高。 | [37] |
3 压电式微喷技术实施细胞打印的优势
压电式微喷技术作为一种非接触式打印方式,经过近几年的发展,已经在组织工程、生物医药领域、柔性可穿戴设备、光学器件等领域得到一定的应用。在细胞打印方面的应用优势,主要体现在如下两大方面:
3.1 高存活率细胞获取
Xu T等[41]于2005年应用微喷射技术实现了哺乳动物仓鼠卵巢细胞打印,且超过90%的细胞能够保持活性,经细胞培养基处理和培养,证明细胞在25天的培养过程中不断增殖;在随后的研究中,又将人类成纤维细胞存活率提高至94%~98%。英国剑桥大学研究小组[42]于2013年使用一种压电喷墨打印头,让成年老鼠的神经胶质细胞(glia cell)和视网膜神经节细胞 (retinal ganglion cells)通过一个不到1毫米的喷嘴“打印”出来,尽管有大量细胞因为沉积在喷墨液底部无法被“打印”出来,造成了损失,但已“打印”出来的细胞都很健康,虽然经过喷嘴喷射,但柔弱的细胞膜仍旧存活着,并能在培养器中生长和保持正常生物特性。Ferris C等[37]以无内毒素低酰基结冷胶悬浮液为生物油墨打印了小鼠成肌细胞,其存活率>95%。2016年以来,Detsch R等[43]分析了压电喷墨参数对结直肠癌上皮细胞打印的影响,并将骨髓基质细胞打印培养,存活率达到 98%;Kim Y K等[14]用不同直径的喷头对老鼠成纤维细胞和人胚肾细胞进行打印,存活率>94%;Ng W L等[16]用50 μm喷头打印新生儿包皮成纤维细胞,存活率达95%。这些研究证明,利用压电式微喷射技术能够进行细胞打印,并且保持较高的细胞存活率。表2列出了几种细胞压电式打印的情况。
表2 几种细胞压电式打印情况
Table 2
| 细胞种类 | 生物油墨 | 打印喷头 | 细胞存活率 | 参考文献 |
|---|---|---|---|---|
| 酵母细胞 | 酵母-麦芽汁溶液 | F181010压电打印头 | 96% | [12] |
| 鼠成纤维细胞 | 胎牛血清-抗生素/抗真菌溶液 | 30μm压电喷头 | 94.4% | [14] |
| 新生儿包皮成纤维细胞 | 聚乙烯吡咯烷酮基混合液 | 50μm压电喷头 | 95% | [16] |
| 小鼠成肌细胞 | 无内毒素低酰基结冷胶悬浮液 | XAAR-126压电喷墨打印头 | 95% | [37] |
| 仓鼠卵巢细胞 | 磷酸盐缓冲液 | 改进的HP51626a压电打印头 | 90% | [41] |
| 骨髓基质细胞 | 纤维蛋白原和凝血酶悬浮液 | 50μm压电喷头 | 98% | [43] |
图5
图6
实验证明,使用压电式微喷射打印技术培养的细胞与传统工艺培养的细胞相比,其最终存活率并没有降低。
3.2 高精度和高效率构建细胞三维组织
图7
图8
Lorber B等[42]及Owens C M等[62]应用压电式喷墨生物打印系统证明了小鼠骨髓干细胞和大鼠视网膜神经节细胞打印构建神经管等神经系统的可行性。Lee V等[63]使用3D喷墨打印技术将胶原蛋白、角化细胞、成纤维细胞分别印刷在不同的结构层,创建了一个具有真皮和表皮结构的模拟皮肤组织,然后将打印的皮肤放置在气液界面培养2周,发现角化细胞正常分化形成多层表皮角质层。Xu T等[64]以含有兔软骨细胞弹性纤维蛋白和胶原蛋白的混合水凝胶作为生物油墨,通过混合喷墨打印系统制备1 mm厚的组织工程软骨支架来满足软骨组织的生物力学性能,然后在体外培养1周后发现水凝胶支架中超过80%的软骨细胞仍保留增殖活性,生物力学检测发现软骨支架的机械性能有明显增强,具有良好的生物学性能。
由于压电式微喷射打印的液滴精度、高喷射速率以及可多通道打印的特点,使其不仅能实现单细胞打印,还可以同时打印多种细胞、细胞外基质和生物材料,并在高通量的细胞排列或繁重复杂的组织结构打印方面有着潜在的优势,为器官制造移植提供有力保障。
4 总 结
压电式微喷技术高精度、高效率、非接触式和低成本的优点,使其在生物细胞打印领域应用关注度越来越高,并具有十分广泛的发展前景。目前,微喷射打印在细胞培养和组织工程领域已经开展诸多拓展,不仅仅局限于简单的三维构架,更对细胞的培养、组织器官多样细胞的打印构建、生物传感器打印、DNA合成等方面提出了更高的要求。由此提出的问题是如何进一步提高细胞的存活率、如何保证细胞打印前后的培养环境、如何精确控制打印参数与生物油墨对细胞打印的影响?因此,微量喷射技术在细胞打印领域的研究以及复杂结构前期设计和后期处理都是需要发展创新的部分。相信在不久的将来,随着人工智能和材料科学的不断进步,生物打印组织器官能够利用在生物细胞打印领域愈发成熟的微喷射技术而实现突破。
参考文献
应用3D打印技术制作组织工程支架:修复骨缺损的研究回顾
.
Tissue-engineered scaffold preparation using three-dimensional printing technology:a retrospective study on bone repair
.
3D打印骨组织工程支架的研究与应用
.
3D printing of bone tissue engineering scaffolds
.
3D生物打印在组织/器官类似物制造领域的应用
.
Three-dimensional bioprinting of tissue/organ analogues:a review on techniques,materials and processes
.
生物3D 打印在神经科学领域的最新进展
.
The latest advances of biological 3D printing in the field of neuroscience
.
3D打印技术在植入式医疗器械中的应用
.
3D Printing:The application in medicine devices
.
生物细胞三维打印技术与材料研究进展
.
Research progress on the technique and materials for three-dimensional bio-printing
.
Three-dimensional bio-printing
.
3D打印技术在细胞打印方面的应用与发展
.
Application and development of 3D printing technology in cell printing
.
细胞打印技术及应用
.
The application of cell bioprinting
Am icroscale in vitro physiolog icalmodel of the liver:predictive screen s for drug metabolism and enzyme induction
.In vitro models of the liver using isolated primary hepatocytes have been used as screens for measuring the metabolism, toxicity and efficacy of xenobiotics, for studying hepatocyte proliferation, and as bioartificial liver support systems. Yet, primary isolated hepatocytes rapidly lose liver specific functions when maintained under standard in vitro cell culture conditions. Many modifications to conventional culture methods have been developed to foster retention of hepatocyte function. Still, not all of the important functions -- especially the biotransformation functions of the liver -- can as yet be replicated at desired levels, prompting continued development of new culture systems. In the first part of this article, we review primary hepatocyte in vitro systems used in metabolism and enzyme induction studies. We then describe a scalable microreactor system that fosters development of 3D-perfused micro-tissue units and show that primary rat cells cultured in this system are substantially closer to native liver compared to cells cultured by other in vitro methods, as assessed by a broad spectrum of gene expression, protein expression and biochemical activity metrics. These results provide a foundation for extension of this culture model to other applications in drug discovery - as a model to study drug-drug interactions, as a model for the assessment of acute and chronic liver toxicity arising from exposure to drugs or environmental agents; and as a disease model for the study of viral hepatitis infection and cancer metastasis.
3D生物打印在再生医学中的应用及展望
.
Application and prospects of 3D bioprintnig in regenerative medicine
细胞打印过程中的细胞受损分析
.
Cell damage analysis in cell printing
Study of droplet formation process during drop-on-demand inkjetting of living cell-laden bioink
.Abstract Biofabrication offers a great potential for the fabrication of three-dimensional living tissues and organs by precisely layer-by-layer placing various tissue spheroids as anatomically designed. Inkjet printing of living cell-laden bioink is one of the most promising technologies enabling biofabrication, and the bioink printability must be carefully examined for it to be a viable biofabrication technology. In this study, the cell-laden bioink droplet formation process has been studied in terms of the breakup time, droplet size and velocity, and satellite formation using a time-resolved imaging approach. The bioink has been prepared using fibroblasts and sodium alginate with four different cell concentrations: without cells, 1 0103 10(6), 5 0103 10(6), and 1 0103 10(7) cells/mL to appreciate the effect of cell concentration on the droplet formation process. Furthermore, the bioink droplet formation process is compared with that during the inkjetting of a comparable polystyrene microbead-laden suspension under the identical operating conditions to understand the effect of particle physical properties on the droplet formation process. It is found that (1) as the cell concentration of bioink increases, the droplet size and velocity decrease, the formation of satellite droplets is suppressed, and the breakup time increases, and (2) compared to the hard bead-laden suspension, the bioink tends to have a less ejected fluid volume, lower droplet velocity, and longer breakup time.
Drop-on-demand inkjet-based cell printing with 30-μm nozzle diameter for cell-level accuracy
.Abstract We present drop-on-demand inkjet-based mammalian cell printing with a 30-μm nozzle diameter for cell-level accuracy. High-speed imaging techniques have been used to analyze the go-and-stop movement of cells inside the nozzle under a pulsed pressure generated by a piezo-actuator and the jet formation after ejection. Patterning of an array of 20 × 20 dots on a glass substrate reveals that each printed drop contains 1.30 cells on average at the cell concentration of 5.0 × 1068cells ml6301 for the very small nozzle, whereas larger nozzles with the diameter of 50 and 80 μm deliver 2.57 and 2.88 cells per drop, respectively. The effects of the size and concentration of printed cells on the number of cells have also been investigated. Furthermore, the effect of the nozzle diameter on printed cells has been evaluated through an examination of viability, proliferation, and morphology of cells by using a live/dead assay kit, CCK-8 assay, and cellular morphology imaging, respectively. We believe that the 30-μm inkjet nozzle can be used for precise cell deposition without any damages to the printed mammalian cells.
Piezoelectric inkjet printing of polymers:stem cell patterning on polymer substrates
.Generating patterns of cells on surfaces is of great significance not only for fundamental studies in biomedical science but also for the creation of functional customized tissue or organs in regenerative medicine. In this paper, arbitrary, complex stem cell patterns were created using piezoelectric inkjet printing of biocompatible polymers. After a systematic study with different inkjet process variables, various poly(lactic-co-glycolic acid) (PLGA) patterns were fabricated on a polystyrene (PS) substrate. Human adipose-derived stem cells (hASCs) were isolated from subcutaneous adipose tissue and were seeded on the PLGA-patterned PS substrate which consists of areas either favorable (PLGA) or unfavorable (bare PS) to cell adhesion. The hASC stably attached, proliferated within the PLGA patterns, thus, complex and confluent hASC patterns were created. Polymer micro-patterning by inkjet printing could be an effective technique to control cell adhesion geometry, leading to arbitrary cell patterning on surfaces.
Polyvinylpyrrolidone-based bio-ink improves cell viability and homogeneity during drop-on-demand printing
.Drop-on-demand (DOD) bioprinting has attracted huge attention for numerous biological applications due to its precise control over material volume and deposition pattern in a contactless printing approach. 3D bioprinting is still an emerging field and more work is required to improve the viability and homogeneity of printed cells during the printing process. Here, a general purpose bio-ink was developed using polyvinylpyrrolidone (PVP) macromolecules. Different PVP-based bio-inks (0%鈥3% w/v) were prepared and evaluated for their printability; the short-term and long-term viability of the printed cells were first investigated. The Z value of a bio-ink determines its printability; it is the inverse of the Ohnesorge number (Oh), which is the ratio between the Reynolds number and a square root of the Weber number, and is independent of the bio-ink velocity. The viability of printed cells is dependent on the Z values of the bio-inks; the results indicated that the cells can be printed without any significant impairment using a bio-ink with a threshold Z value of 9.30 (2% and 2.5% w/v). Next, the cell output was evaluated over a period of 30 min. The results indicated that PVP molecules mitigate the cell adhesion and sedimentation during the printing process; the 2.5% w/v PVP bio-ink demonstrated the most consistent cell output over a period of 30 min. Hence, PVP macromolecules can play a critical role in improving the cell viability and homogeneity during the bioprinting process.
Microfluidic 3D bone tissue model for high-throughput evaluation of wound-healing and infection-preventing biomaterials
.
URL
PMID:22061488
Magsci
[本文引用: 1]

We report the use of a microfluidic 3D bone tissue model, as a high-throughput means of evaluating the efficacy of biomaterials aimed at accelerating orthopaedic implant-related wound-healing while preventing bacterial infection. As an example of such biomaterials, inkjet-printed micropatterns were prepared to contain antibiotic and biphasic calcium phosphate (BCP) nanoparticles dispersed in a poly(d,l-lactic-co-glycolic) acid matrix. The micropatterns were integrated with a microfluidic device consisting of eight culture chambers. The micropatterns immediately and completely killed Staphylococcus epidermidis upon inoculation, and enhanced the calcified extracellular matrix production of osteoblasts. Without antibiotic elution, bacteria rapidly proliferated to result in an acidic microenvironment which was detrimental to osteoblasts. These results were used to demonstrate the tissue model potential in: (i) significantly reducing the number of biomaterial samples and culture experiments required to assess invitro efficacy for wound-healing and infection prevention and (ii) in situ monitoring of dynamic interactions of biomaterials with bacteria as wells as with tissue cells simultaneously.
A novel approach for precisely controlled multiple cell patterning in microfluidic chips by inkjet printing and the detection of drug metabolism and diffusion
.In this work we report the use of inkjet printing as a precise and convenient means for microscale cell patterning in microfluidic chips followed by cell co-culture, stimulation and analysis. A self-made inkjet printing device was manufactured with adjustable parameters, which was capable of multiple cell printing within biocompatible materials. Sodium alginate was used as a printing matrix for cell encapsulation, and precisely distributed cell arrays on glass slides were obtained by accurate software controlled printing. By covering a PDMS layer with the corresponding microchannels onto the cell array substrate and subsequently injecting an ion cross-linking reagent, the cells containing alginate arrays gelated immediately and were immobilized on the bottom of the microchip, which could be utilized for cell culture and analysis. HepG2 cells and U251 cells were successfully co-patterned in the microchip and used for drug metabolism and diffusion experiment to imitate the in vivo situation, as a means to ascertain the capability of the system for precise microscale cell patterning in a microchip. The prodrug tegafur was metabolized by HepG2 cells into the active anticancer compound 5-fluorouracil and this produced an adverse gradient effect on U251 cells according to the distance from the HepG2 cells. The developed approach presented a feasible way to integrate inkjet cell printing and microfluidic chips for the first time, which is proved to be capable of spatially controlled printing of multiple kinds of cells into a microchip for cell culture, stimulation and analysis, which could be applied to tissue engineering, drug testing and related areas. We envision that the approach will help significantly increase the cell patterning efficacy in microfluidic chips as well as reduce the extent of laborious experimental work.
Piezoelectric inkjet-based single-cells printing by image processing for high efficiency and automatic cell printing//17th International Conference on
ABSTRACTResearch in cell printing by piezoelectric inkjet printer has been conducted, however, relatively reliable single-cells printing yield had not been obtained. We proposed a different approach to increase the yield in single-cells printing by utilizing piezoelectric driving method not used in recent inkjet printer devices, push-pull method. Furthermore, cell recognition system constructed by image processing algorithm was used to predict whether individual cell is present at the tip of inkjet head. Combining both methods, we successfully developed a single-cells printing system by piezoelectric inkjet printer with single-cells printing yield of up to 98%.
Cell patterning through inkjet printing of one cell per droplet
.The inkjet ejection technology used in printers has been adopted and research has been conducted on manufacturing artificial tissue by patterning cells through micronozzle ejection of small droplets containing multiple cells. However, stable injection of cells has proven difficult, owing to the frequent occurrence of nozzle clogging. In this paper, a piezoelectric inkjet head constructed with a glass capillary that enabled viewing of the nozzle section was developed, the movement of cells ejected from the nozzle tip was analyzed, and a method for stably ejecting cells was verified. A pull-push ejection method was compared with a push-pull ejection method regarding the voltage waveform applied to the piezoelectric element of the head. The push-pull method was found to be more suitable for stable ejection. Further, ejection of one cell per droplet was realized by detecting the position of the cell in the nozzle section and utilizing these position data. Thus, a method for more precise patterning of viable cells at desired position and number was established. This method is very useful and promising not only for biofabrication, 3D tissue construction, cell printing, but also for a number of biomedical application, such as bioMEMS, lab on a chip research field.
1-x)Bi0.5Na0.5TiO3-TiO(3)lead-free piezoelectric ceramics for energy-harvesting applications
.Lead-free (1 - x)Bi0.5Na0.5TiO3-xBaTiO(3) piezoelectric ceramics were investigated for energy-harvesting applications. In order to enhance the piezoelectric properties, the BaTiO3 content was varied at 0 <= x <= 0.1. Furthermore, the sintering temperature was optimized to enhance the piezoelectric properties and output energies. The piezoelectric charge coefficient was enhanced from 70 to 164 pC/N by introducing BaTiO3 and optimizing the sintering temperature to 1175 degrees C. The (1 - x)Bi0.5Na0.5TiO3-xBaTiO(3) lead-free piezoelectric ceramics exhibited a piezoelectric voltage coefficient of 47.03 x 10(-3) V m/N at x=0.04, which is slightly higher than the piezoelectric voltage coefficient of lead-based piezoelectric ceramics. The generated output voltage and output power were 8.95 V and 18 nW, respectively, for 0.94Bi(0.5)Na(0.5)TiO(3)-0.06BaTiO(3) ceramics sintered at 1175 degrees C. The results suggest that lead-free (1 -x)Bi0.5Na0.5TiO3-xBaTiO(3) piezoelectric ceramics are good candidates for energy-harvesting applications. (C) 2015 Elsevier Ltd. All rights reserved.
Study of living cell distribution during inkjet printing of bioink//Proceedings of the asme international manufacturing science and engineering conference 2017.Los Angeles,
Delivery of human fibroblast cells by piezoelectric drop-on-demand inkjet printing
.A piezoelectric actuated, drop-on-demand inkjet printing system has been used to deliver suspensions of human fibroblast cells from a well-characterized cell line (HT 1080) in order to investigate the behaviour of cells exposed to the mechanical and fluid stresses associated with the printing process. By varying the amplitude and rise time of the electrical pulse used to excite the piezoelectric actuator, it is possible to alter the stresses experienced by the cells. It is shown that the amplitude of the pulse has a small influence on cell survivability with regression analysis showing cell survival rates falling from 98% with a 40 V pulse (indistinguishable from control measurements) to approximately 94% with a 80 V pulse. The rise time of the pulse was found to have no influence on cell survival. Cell viability post-printing was also assessed using the Alamar Blue metabolic assay and the cells that survived were unaffected by the printing process, with neither pulse amplitude nor rise time showing any significant influence on cell viability (using the standard 5% probability threshold). However, inkjet printing requires cell suspensions to be stable over several minutes during the printing process and it was found that after about 20 min printing, some cell agglomeration or sedimentation affected the printing performance.
Reduction of droplet volume by controlling actuating waveforms in inkjet printing for micro-pattern formation
.Inkjet printing has proven to be a promising and flexible process methodology for low cost and drop-on-demand pattern formation in small-scale production of micro-electro-mechanical systems. To optimize the micro-patterns formed by inkjet printing, an accurate control of droplet volume is essential and critical. In this study, an inkjet system with a nozzle driven by a circular piezoelectric element was used to explore the impact of different waveforms on droplet volume. The investigation into this study included the impact of unipolar, bipolar, M-shaped and W-shaped waveforms as well as the effects of their amplitudes and pulse durations. The inkjetting behavior of Newtonian and non-Newtonian fluids under different actuating waveforms was studied in order to obtain a maximum reduction in ejected droplet sizes. An effective reduction of droplet volume in the range of 50 80% was demonstrated. The results of inkjetting PEDOT ink on a polished silicon surface showed that a 50% reduction in line width was achieved
A waveform design method for highspeed inkjet printing based on self-sensing measurement
.A high-speed waveform design method for piezo inkjet printing is proposed. This method uses the pressure wave information inside the ink dispenser. The pressure wave inside the inkjet dispenser can be effectively measured from the piezo current because the piezo has the self-sensing capability. The measured pressure wave was compared with the dispenser vibration measured by the laser Doppler vibrometer. For high-speed inkjet printing, a two-pulse waveform was designed based on the measured pressure wave to effectively suppress the residual pressure wave after jetting.
A novel method for fine patterning by piezoelectrically induced pressure adjustment of inkjet printing
.In this paper, a method is proposed for fine line pattern formation on polyimide films by drop-on-demand inkjet printing without the need to reduce the nozzle size. The rapid change in the shape of a piezoelectric actuator caused by a pressure wave causes the fluid to be ejected as droplets. By shortening the duration of the chamber compression period, smaller droplets can be ejected from the nozzle orifice. The pressure wave is a key factor in determination of the line width. A 20- 08 m-wide line pattern pulsed at 0.5 08 s intervals was successfully fabricated using a Cu complex ion ink and a cartridge with volume of 1 pl. This method is expected to be widely used in various applications that demand high-resolution patterning. The 20- 08 m line width of the conducting track is a particularly useful size for fabrication of transparent electrodes.
基于细胞打印的压电式喷墨头研究进展
.
Research progress of piezoelectric inkjet head based on cell printing
.
Numerical-calculation of the fluid-dynamics of drop-on-demand jets
.A numerical method that makes use of the complete incompressible flow equations with a free surface is discussed and used to study an impulsively driven laminar jet. Flow behavior dependence upon fluid properties (characterized by a Reynolds number over Weber number nondimensionalization) is compared for drop integrity purposes. Several variations of square wave pressure history applied at a nozzle inlet are discussed in relation to drop velocities produced and structure of ejected drops. Timewise development of flow both interior and exterior to the nozzle is illustrated through computed contour sequences.
Ink-jet delivery of particle suspensions by piezoelectric droplet ejectors
.The volume and velocity of droplets ejected from a piezoelectric droplet generator, as used in ink-jet printing, have been studied for a range of concentrated suspensions of submicron alumina particles as a function of driving signal voltage, frequency, and peak shape. Drop velocity and volume are found to show a linear relation with driving voltage, but show a more complicated and periodic behavior with changing frequency and peak width. This periodic dependence is shown to be a function of the acoustic properties of the fluid-filled chamber in the droplet generator. However, a simple model considering propagation of pressure waves along the tubular actuator is not consistent with experimental data if the ends are modeled as step changes in acoustic impedance. By comparing the data with that in the literature, it is proposed that the acoustic boundary condition (impedance change) at the orifice where drops are ejected is a function of orifice size and extrapolates to a closed boundary condition, as the orifice diameter tends to zero. At equivalent driving signal frequencies, the drop volume ejected, normalized by actuator volume displacement, is shown to be a function of the Ohnesorge number of the orifice through which the drops are ejected.
Inkjet printing of functional and structural materials:fluid property requirements,feature stability,and resolution
.Inkjet printing is viewed as a versatile manufacturing tool for applications in materials fabrication in addition to its traditional role in graphics output and marking. The unifying feature in all these applications is the dispensing and precise positioning of very small volumes of fluid (1-100 picoliters) on a substrate before transformation to a solid. The application of inkjet printing to the fabrication of structures for structural or functional materials applications requires an understanding as to how the physical processes that operate during inkjet printing interact with the properties of the fluid precursors used. Here we review the current state of understanding of the mechanisms of drop formation and how this defines the fluid properties that are required for a given liquid to be printable. The interactions between individual drops and the substrate as well as between adjacent drops are important in defining the resolution and accuracy of printed objects. Pattern resolution is limited by the extent to which a liquid drop spreads on a substrate and how spreading changes with the overlap of adjacent drops to form continuous features. There are clearly defined upper and lower bounds to the width of a printed continuous line, which can be defined in terms of materials and process variables. Finer-resolution features can be achieved through appropriate patterning and structuring of the substrate prior to printing, which is essential if polymeric semiconducting devices are to be fabricated. Low advancing and receding contact angles promote printed line stability but are also more prone to solute segregation or offee staining on drying.
Influence of fluid physical properties on ink-jet printability
.Ink-jet printing is a method for directly patterning and fabricating patterns without the need for masks.To achieve this,the fluids used as inks must have the capability of being stably and accurately printed by ink-jetting.We have investigated the inter-relationship between ink-jet printability and physical fluid properties by monitoring droplet formation dynamics.The printability of the fluids was determined using the inverse(Z)of the Ohnesorge number(Oh)which relates to the viscosity,surface tension,and density of the fluid.We have experimentally defined the printable range as 4≤Z≤ 14 by considering characteristics such as single droplet formability,positional accuracy,and maximum allowable jettingfrequency.
Improving piezoelectric cell printing accuracy and reliability through neutral buoyancy of suspensions
.The sedimentation and aggregation of cells within inkjet printing systems has been hypothesized to negatively impact printer performance. The purpose of this study was to investigate this influence through the use of neutral buoyancy. Ficoll PM400 was used to create neutrally buoyant MCF-7 breast cancer cell suspensions, which were ejected using a piezoelectric drop-on-demand inkjet printing system. It was found that using a neutrally buoyant suspension greatly increased the reproducibility of consistent cell counts, and eliminated nozzle clogging. Moreover, the use of Ficoll PM400 was shown to not affect cellular viability. This is the first demonstration of such scale and accuracy achieved using a piezoelectric inkjet printing system for cellular dispensing. Biotechnol. Bioeng. 2012; 109: 2932–2940. 08 2012 Wiley Periodicals, Inc.
Statistical Modeling of single target cell encapsulation
.High throughput drop-on-demand systems for separation and encapsulation of individual target cells from heterogeneous mixtures of multiple cell types is an emerging method in biotechnology that has broad applications in tissue engineering and regenerative medicine, genomics, and cryobiology. However, cell encapsulation in droplets is a random process that is hard to control. Statistical models can provide an understanding of the underlying processes and estimation of the relevant parameters, and enable reliable and repeatable control over the encapsulation of cells in droplets during the isolation process with high confidence level. We have modeled and experimentally verified a microdroplet-based cell encapsulation process for various combinations of cell loading and target cell concentrations. Here, we explain theoretically and validate experimentally a model to isolate and pattern single target cells from heterogeneous mixtures without using complex peripheral systems.
One cell-one well:A new approach to inkjet printing single cell microarrays
.Abstract A new approach to prepare arrays of sessile droplets of living single cell cultures using a liquid hydrophobic barrier prevents the samples from dehydrating, and allows for spatially addressable arrays for statistical quantitative single cell studies. By carefully advancing a thin layer of mineral oil on the substrate over the droplets during the printing, dehydration of the droplets can be prevented, and the vitality of the cells can be maintained. The net result of this confluence of submerged cell culturing and inkjet printing is facile access to spatially addressable arrays of isolated single cells on surfaces. Such single cell arrays may be particularly useful as high-throughput tools in the rapidly emerging "omics" fields of cell biology.
Rheological manipulation for improved reliability in inkjet printing of living cells//2016 IEEE 29th International Conference on Micro Electro Mechanical Systems.Shanghai,
Investifation of the hydrodynamics of suspended cells for reliable inkjet cell printing /ASME 2014 12th International Conference on Nanochannels,Microchannels,and Minichannels.Chicago,
Bio-ink for on-demand printing of living cells
.Drop-on-demand bioprinting allows the controlled placement of living cells, and will benefit research in the fields of tissue engineering, drug screening and toxicology. We show that a bio-ink based on a novel microgel suspension in a surfactant-containing tissue culture medium can be used to reproducibly print several different cell types, from two different commercially available drop-on-demand printing systems, over long printing periods. The bio-ink maintains a stable cell suspension, preventing the settling and aggregation of cells that usually impedes cell printing, whilst meeting the stringent fluid property requirements needed to enable printing even from many-nozzle commercial inkjet print heads. This innovation in printing technology may pave the way for the biofabrication of multi-cellular structures and functional tissue.
Cell damage evaluation of thermal inkjet printed chinese hamster ovary cells
.Thermal inkjet printing technology has been applied successfully to cell printing. However, there are concerns that printing process may cause cell damages or death. We conducted a comprehensive study of thermal inkjet printed Chinese hamster ovary (CHO) cells by evaluating cell viability and apoptosis, and possible cell membrane damages. Additionally, we studied the cell concentration of bio-ink and found optimum printing of concentrations around 8 million cells per mL. Printed cell viability was 89% and only 3.5% apoptotic cells were observed after printing. Transient pores were developed in the cell membrane of printed cells. Cells were able to repair these pores within 265h after printing. Green fluorescent protein (GFP) DNA plasmids were delivered to CHO-S cells by co-printing. The transfection efficiency is above 30%. We conclude that thermal inkjet printing technology can be used for precise cell seeding with minor effects and damages to the printed mammalian cells. The printing process causes transient pores in cell membranes, a process which has promising applications for gene and macroparticles delivery to induce the biocompatibility or growth of engineered tissues. Biotechnol. Bioeng. 2010;106: 963–969. 08 2010 Wiley Periodicals, Inc.
Bioprinting toward organ fabrication:Challenges and future trends
.
URL
PMID:23372076
Magsci
[本文引用: 1]

Tissue engineering has been a promising field of research, offering hope for bridging the gap between organ shortage and transplantation needs. However, building three-dimensional (3-D) vascularized organs remains the main technological barrier to be overcome. Organ printing, which is defined as computer-aided additive biofabrication of 3-D cellular tissue constructs, has shed light on advancing this field into a new era. Organ printing takes advantage of rapid prototyping (RP) technology to print cells, biomaterials, and cell-laden biomaterials individually or in tandem, layer by layer, directly creating 3-D tissue-like structures. Here, we overview RP-based bioprinting approaches and discuss the current challenges and trends toward fabricating living organs for transplant in the near future.
Cell and organ printing turns 15:Diverse research to commercial transtitions
.
Inkjet printing of viable mammalian cells
.
URL
PMID:15193884
Magsci
[本文引用: 1]

The purpose of this study was to explore the use of a commercial thermal printer to deposit Chinese Hamster Ovary (CHO) and embryonic motoneuron cells into pre-defined patterns. These experiments were undertaken to verify the biocompatibility of thermal inkjet printing of mammalian cells and the ability to assemble them into viable constructs. Using a modified Hewlett Packard (HP) 550C computer printer and an HP 51626a ink cartridge, CHO cells and rat embryonic motoneurons were suspended separately in a concentrated phosphate buffered saline solution (3×). The cells were subsequently printed as a kind of “ink” onto several “bio-papers” made from soy agar and collagen gel. The appearance of the CHO cells and motoneurons on the bio-papers indicated an healthy cell morphology. Furthermore, the analyses of the CHO cell viability showed that less than 8% of the cells were lysed during printing. These data indicate that mammalian cells can be effectively delivered by a modified thermal inkjet printer onto biological substrates and that they retain their ability to function. The computer-aided inkjet printing of viable mammalian cells holds potential for creating living tissue analogs, and may eventually lead to the construction of engineered human organs.
Adult rat retinal ganglion cells and glia can be printed by piezoelectric injet printing
.Abstract We have investigated whether inkjet printing technology can be extended to print cells of the adult rat central nervous system (CNS), retinal ganglion cells (RGC) and glia, and the effects on survival and growth of these cells in culture, which is an important step in the development of tissue grafts for regenerative medicine, and may aid in the cure of blindness. We observed that RGC and glia can be successfully printed using a piezoelectric printer. Whilst inkjet printing reduced the cell population due to sedimentation within the printing system, imaging of the printhead nozzle, which is the area where the cells experience the greatest shear stress and rate, confirmed that there was no evidence of destruction or even significant distortion of the cells during jet ejection and drop formation. Importantly, the viability of the cells was not affected by the printing process. When we cultured the same number of printed and non-printed RGC/glial cells, there was no significant difference in cell survival and RGC neurite outgrowth. In addition, use of a glial substrate significantly increased RGC neurite outgrowth, and this effect was retained when the cells had been printed. In conclusion, printing of RGC and glia using a piezoelectric printhead does not adversely affect viability and survival/growth of the cells in culture. Importantly, printed glial cells retain their growth-promoting properties when used as a substrate, opening new avenues for printed CNS grafts in regenerative medicine.
Evaluation of cell inkjet printing technique for biofabrication
.
Emerging technologies in medical applications of minimum volume vitrification
.Cell/tissue biopreservation has broad public health and socio-economic impact affecting millions of lives. Cryopreservation technologies provide an efficient way to preserve cells and tissues targeting the clinic for applications including reproductive medicine and organ transplantation. Among these technologies, vitrification has displayed significant improvement in post-thaw cell viability and function by eliminating harmful effects of ice crystal formation compared to the traditional slow freezing methods. However, high cryoprotectant agent concentrations are required, which induces toxicity and osmotic stress to cells and tissues. It has been shown that vitrification using small sample volumes (i.e., <1 碌l) significantly increases cooling rates and hence reduces the required cryoprotectant agent levels. Recently, emerging nano- and micro-scale technologies have shown potential to manipulate picoliter to nanoliter sample sizes. Therefore, the synergistic integration of nanoscale technologies with cryogenics has the potential to improve biopreservation methods.
Assessment of tumourigenic potential in long-term cryopreserved human adipose-derived stem cells
.Abstract Cryopreservation represents an efficient way to preserve human mesenchymal stem cells (hMSCs) at early culture/passage, and allows pooling of cells to achieve sufficient cells required for off-the-shelf use in clinical applications, e.g. cell-based therapies and regenerative medicine. To fully apply cryopreserved hMSCs in a clinical setting, it is necessary to evaluate their biosafety, e.g. chromosomal abnormality and tumourigenic potential. To date, many studies have demonstrated that cryopreserved hMSCs display no chromosomal abnormalities. However, the tumourigenic potential of cryopreserved hMSCs has not yet been evaluated. In the present study, we cryopreserved human adipose-derived mesenchymal stem cells (hASCs) for 3onths, using a slow freezing method with various cryoprotective agents (CPAs), followed by assessment of the tumourigenic potential of the cryopreserved hASCs after thawing and subculture. We found that long-term cryopreserved hASCs maintained normal levels of the tumour suppressor markers p53, p21, p16 and pRb, hTERT, telomerase activity and telomere length. Further, we did not observe significant DNA damage or signs of p53 mutation in cryopreserved hASCs. Our findings suggest that long-term cryopreserved hASCs are at low risk of tumourigenesis. These findings aid in establishing the biosafety profile of cryopreserved hASCs, and thus establishing low hazardous risk perception with the use of long-term cryopreserved hASCs for future clinical applications. Copyright 2016 John Wiley & Sons, Ltd.
High through put cryopreservation of cells by rapid freezing of sub-μl drops using inkjet printing-cryoprinting
.
Trends in risks associated with new drug development:Success rates for investigational drugs
.This study utilizes both public and private data sources to estimate clinical phase transition and clinical approval probabilities for drugs in the development pipelines of the 50 largest pharmaceutical firms (by sales). The study examined the development histories of these investigational compounds from the time point at which they first entered clinical testing (1993-2004) through June 2009. The clinical approval success rate in the United States was 16% for self-originated drugs (originating from the pharmaceutical company itself) during both the 1993-1998 and the 1999-2004 subperiods. For all compounds (including licensed-in and licensed-out drugs in addition to self-originated drugs), the clinical approval success rate for the entire study period was 19%. The estimated clinical approval success rates and phase transition probabilities differed significantly by therapeutic class. The estimated clinical approval success rate for self-originated compounds over the entire study period was 32% for large molecules and 13% for small molecules. The estimated transition probabilities were also higher for all clinical phases with respect to large molecules.
Clinical development success rates for investigational drugs
.
URL
PMID:24406927
Magsci
[本文引用: 1]

Elsevier’s Scopus, the largest abstract and citation database of peer-reviewed literature. Search and access research from the science, technology, medicine, social sciences and arts and humanities fields.
Fabrication of in vitro cancer microtissue array on fibroblast-layered nanofibrous membrane by inkjet printing
.In general, a drug candidate is evaluated using 2D-cultured cancer cells followed by an animal model. Despite successful preclinical testing, however, most drugs that enter human clinical trials fail. The high failure rates are mainly caused by incompatibility between the responses of the current models and humans. Here, we fabricated a cancer microtissue array in a multi-well format that exhibits heterogeneous and batch-to-batch structure by continuous deposition of collagen-suspended Hela cells on a fibroblast-layered nanofibrous membrane via inkjet printing. Expression of both Matrix Metalloproteinase 2 (MMP2) and Matrix Metalloproteinase 9 (MMP9) was higher in cancer microtissues than in fibroblast-free microtissues. The fabricated microtissues were treated with an anticancer drug, and high drug resistance to doxorubicin occurred in cancer microtissues but not in fibroblast-free microtissues. These results introduce an inkjet printing fabrication method for cancer microtissue arrays, which can be used for various applications such as early drug screening and gradual 3D cancer studies.
Direct freeform fabrication of seeded hydrogels in arbitrary geometries
.
Three-dimensional tissue constructs built by bioprinting
.Bioprinting is an evolving tissue engineering technology. It utilizes computer controlled three-dimensional printers for rapid and high-precision construction of three-dimensional biological structures. We employed discrete and continuous bioprinting to build three-dimensional tissue constructs. In the former case bioink particles - spherical cell aggregates composed of many thousands of cells - are delivered one by one into biocompatible scaffolds, the biopaper. Structure formation takes place by the subsequent fusion of the bioink particles due to their liquid-like and self-assembly properties. In the latter case a mixture of cells and scaffold material is extruded from the biocartridge akin to toothpaste to arrive at the desired construct. Specifically, we built rectangular tissue blocks of several hundred microns in thickness as well as tubular structures of several millimeters in height. The physical basis of structure formation was studied by computer simulations.
Current advances and future perspectives in extrusion-based bioprinting
.Extrusion-based bioprinting (EBB) is a rapidly growing technology that has made substantial progress during the last decade. It has great versatility in printing various biologics, including cells, tissues, tissue constructs, organ modules and microfluidic devices, in applications from basic research and pharmaceutics to clinics. Despite the great benefits and flexibility in printing a wide range of bioinks, including tissue spheroids, tissue strands, cell pellets, decellularized matrix components, micro-carriers and cell-laden hydrogels, the technology currently faces several limitations and challenges. These include impediments to organ fabrication, the limited resolution of printed features, the need for advanced bioprinting solutions to transition the technology bench to bedside, the necessity of new bioink development for rapid, safe and sustainable delivery of cells in a biomimetically organized microenvironment, and regulatory concerns to transform the technology into a product. This paper, presenting a first-time comprehensive review of EBB, discusses the current advancements in EBB technology and highlights future directions to transform the technology to generate viable end products for tissue engineering and regenerative medicine.
Biofabrication of multi-material anatomically shaped tissue constructs
.
Bioprinting of human pluripotent stem cells and their directed differentiation into hepatocyte-like cells for generation of mini-livers in 3D
.We report the first investigation into the bioprinting of human induced pluripotent stem cells (hiPSCs), their response to a valve-based printing process as well as their post-printing differentiation into hepatocyte-like cells (HLCs). HLCs differentiated from both hiPSCs and human embryonic stem cells (hESCs) sources were bioprinted and examined for the presence of hepatic markers to further validate the compatibility of the valve-based bioprinting process with fragile cell transfer. Examined cells were positive for nuclear factor 4 alpha and were demonstrated to secrete albumin and have morphology that was also found to be similar to that of hepatocytes. Both hESC and hiPSC lines were tested for post-printing viability and pluripotency and were found to have negligible difference in terms of viability and pluripotency between the printed and non-printed cells. hESC-derived HLCs were 3D printed using alginate hydrogel matrix and tested for viability and albumin secretion during the remaining differentiation and were found to be hepatic in nature. 3D printed with 40-layer of HLC-containing alginate structures reached peak albumin secretion at day 21 of the differentiation protocol. This work demonstrates that the valve-based printing process is gentle enough to print human pluripotent stem cells (hPSCs) (both hESCs and hiPSCs) while either maintaining their pluripotency or directing their differentiation into specific lineages. The ability to bioprint hPSCs will pave the way for producing organs or tissues on demand from patient specific cells which could be used for animal-free drug development and personalized medicine.
Sacrificial-layer free transfer of mammalian cells using near infrared femtosecond laser pulses
.Abstract Laser-induced cell transfer has been developed in recent years for the flexible and gentle printing of cells. Because of the high transfer rates and the superior cell survival rates, this technique has great potential for tissue engineering applications. However, the fact that material from an inorganic sacrificial layer, which is required for laser energy absorption, is usually transferred to the printed target structure, constitutes a major drawback of laser based cell printing. Therefore alternative approaches using deep UV laser sources and protein based acceptor films for energy absorption, have been introduced. Nevertheless, deep UV radiation can introduce DNA double strand breaks, thereby imposing the risk of carcinogenesis. Here we present a method for the laser-induced transfer of hydrogels and mammalian cells, which neither requires any sacrificial material for energy absorption, nor the use of UV lasers. Instead, we focus a near infrared femtosecond (fs) laser pulse (脦禄 = 1030 nm, 450 fs) directly underneath a thin cell layer, suspended on top of a hydrogel reservoir, to induce a rapidly expanding cavitation bubble in the gel, which generates a jet of material, transferring cells and hydrogel from the gel/cell reservoir to an acceptor stage. By controlling laser pulse energy, well-defined cell-laden droplets can be transferred with high spatial resolution. The transferred human (SCP1) and murine (B16F1) cells show high survival rates, and good cell viability. Time laps microscopy reveals unaffected cell behavior including normal cell proliferation.
Laser-based 3D cell printing for tissue engineering
.
Study of gelatin as an effective energy absorbing layer for laser bioprinting
.Abstract Laser-induced forward transfer printing, also commonly known as laser printing, has been widely implemented for three-dimensional bioprinting due to its unique orifice-free nature during printing. However, the printing quality has the potential to be further improved for various laser bioprinting applications. The objectives of this study are to investigate the feasibility of using gelatin as an energy absorbing layer (EAL) material for laser bioprinting and its effects on the quality of printed constructs, bioink printability, and post-printing cell viability and process-induced DNA damage. The gelatin EAL is applied between the quartz support and the coating of build material, which is to be printed. Printing quality can be improved by EAL-assisted laser printing when using various alginate solutions (1%, 2%, and 4%) and cell-laden bioinks (2% alginate and 5 × 1068 cells ml6301 in cell culture medium). The required laser fluence is also reduced due to a higher absorption coefficient of gelatin gel, in particular when to achieve the best printing type/quality. The post-printing cell viability is improved by ~10% and DNA double-strand breaks are reduced by ~50%. For all the build materials investigated, the gelatin EAL helps reduce the droplet size and average jet velocity.
3D printing of biomimetic microstructures for cancer cell migration
.Abstract To understand the physical behavior and migration of cancer cells, a 3D in vitro micro-chip in hydrogel was created using 3D projection printing. The micro-chip has a honeycomb branched structure, aiming to mimic 3D vascular morphology to test, monitor, and analyze differences in the behavior of cancer cells (i.e. HeLa) vs. non-cancerous cell lines (i.e. 10 T1/2). The 3D Projection Printing system can fabricate complex structures in seconds from user-created designs. The fabricated microstructures have three different channel widths of 25, 45, and 120 microns wide to reflect a range of blood vessel diameters. HeLa and 10 T1/2 cells seeded within the micro-chip were then analyzed for morphology and cell migration speed. 10 T1/2 cells exhibited greater changes in morphology due to channel size width than HeLa cells; however, channel width had a limited effect on 10 T1/2 cell migration while HeLa cancer cell migration increased as channel width decreased. This physiologically relevant 3D cancer tissue model has the potential to be a powerful tool for future drug discoveries and cancer migration studies.
Accelerated myotube formation using bioprinting technology for biosensor applications
.
URL
PMID:23160742
Magsci
[本文引用: 1]

Muscle-powered, biological, microelectro-mechanical system is promising for actuator and biosensor applications. Functional conjugation between the cells, tissues, and biomolecules to the microdevice is crucial for this application. Bioprinting as an enabling technology possesses the advantages of high throughput, digital control, and highly accurate delivery of various biological factors to the desired locations for numerous applications such as 3D tissue fabrication. We have now evaluated the feasibility of the precise placement of mouse myoblasts onto micro-sized cantilevers. The evenly aligned printed cells fused with each other and formed mature myotubes after only 4 days. In contrast, it took more than 14 days for randomly deposited cells to do so. The printed myotubes were functional and responded to the electrical stimulation synchronously. Furthermore, the integrated Bio-MEMS device responded to the chemical stimulation spontaneously which demonstrated the potential as a functional biosensor. The contractility of the system was recovered quickly after the removal of the chemical stimulation, which indicated the flexibility of this system and the recycling potential.
Freeform inkjet printing of cellular structures with bifurcations
.ABSTRACT Organ printing offers a great potential for the freeform layer-by-layer fabrication of three-dimensional (3D) living organs using cellular spheroids or bioinks as building blocks. Vascularization is often identified as a main technological barrier for building 3D organs. As such, the fabrication of 3D biological vascular trees is of great importance for the overall feasibility of the envisioned organ printing approach. In this study, vascular-like cellular structures are fabricated using a liquid support-based inkjet printing approach, which utilizes a calcium chloride solution as both a cross-linking agent and support material. This solution enables the freeform printing of spanning and overhang features by providing a buoyant force. A heuristic approach is implemented to compensate for the axially-varying deformation of horizontal tubular structures to achieve a uniform diameter along their axial directions. Vascular-like structures with both horizontal and vertical bifurcations have been successfully printed from sodium alginate only as well as mouse fibroblast-based alginate bioinks. The post-printing fibroblast cell viability of printed cellular tubes was found to be above 90% even after a 2465h incubation, considering the control effect. Biotechnol. Bioeng. 2015;112: 1047–1055. 08 2014 Wiley Periodicals, Inc.
Inkjet printing of silk nest arrays for cell hosting
.
URL
PMID:24605757
Magsci
[本文引用: 1]

An inkjet printing approach is presented for the facile fabrication of microscopic arrays of biocompatible silk "nests" capable of hosting live cells for prospective biosensors. The patterning of silk fibroin nests were constructed by the layer-by-layer (LbL) assembly of silk polyelectrolytes chemically modified with poly-(l-lysine) and poly-(l-glutamic acid) side chains. The inkjet-printed silk circular regions with a characteristic "nest" shape had diameters of 70-100 渭m and a thickness several hundred nanometers were stabilized by ionic pairing and by the formation of the silk II crystalline secondary structure. These "locked-in" silk nests remained anchored to the substrate during incubation in cell growth media to provide a biotemplated platform for printing-in, immobilization, encapsulation and growth of cells. The process of inkjet-assisted printing is versatile and can be applied on any type of substrate, including rigid and flexible, with scalability and facile formation.
Biofabrication and testing of a fully cellular nerve graft
.Abstract Rupture of a nerve is a debilitating injury with devastating consequences for the individual's quality of life. The gold standard of repair is the use of an autologous graft to bridge the severed nerve ends. Such repair however involves risks due to secondary surgery at the donor site and may result in morbidity and infection. Thus the clinical approach to repair often involves non-cellular solutions, grafts composed of synthetic or natural materials. Here we report on a novel approach to biofabricate fully biological grafts composed exclusively of cells and cell secreted material. To reproducibly and reliably build such grafts of composite geometry we use bioprinting. We test our grafts in a rat sciatic nerve injury model for both motor and sensory function. In particular we compare the regenerative capacity of the biofabricated grafts with that of autologous grafts and grafts made of hollow collagen tubes by measuring the compound action potential (for motor function) and the change in mean arterial blood pressure as consequence of electrically eliciting the somatic pressor reflex. Our results provide evidence that bioprinting is a promising approach to nerve graft fabrication and as a consequence to nerve regeneration.
Design and fabrication of human skin by three-dimensional bioprinting
.