生殖系统的发育对生物的世代交替以及遗传重组具有重要的意义,植物与动物之间的一个较大的差异便是生殖器官起始发育的时间不同 [1]。多细胞动物的原始生殖细胞形成于胚胎发育时期,而高等植物的早期发育过程中仅形成根、茎、叶等营养器官,随后在由营养生长到生殖生长的转化过程中,由亚表皮层细胞逐渐分化形成花原基等生殖器官 [2,3,4]。生殖器官中大部分细胞为体细胞,仅有少部分细胞分化形成生殖细胞 [5],花药原基中的生殖细胞再经过复杂的分化过程形成孢子母细胞,随后孢子母细胞经过一次减数分裂和两次有丝分裂形成小孢子,完成由多倍体世代到单倍体世代的转化。这个复杂的生物学过程需要孢子体基因与配子体基因的协同调控 [6,7,8],参与此过程的任何基因突变都有可能对植物的育性产生影响,导致雄性不育现象的发生。
植物雄性不育是指在高等植物中,雄性生殖器官不能产生有功能的雄配子,而雌性营养器官及雌性生殖器官发育正常,可受精结实并将不育性遗传给后代的现象 [9]。雄性不育现象于19世纪被发现,并且在高等植物中普遍存在。早在1921年,Eyster就在玉米中发现了雄性不育突变体,随后大量的玉米雄性不育材料被发现或创制,目前MaizeGDB种质资源库中已收集近百份玉米雄性不育材料(http://www.maizegdb.org/data_center/phenotype?id=24992)。作为常异花授粉植物,玉米是三大粮食作物中利用杂种优势起始最早且最为彻底的植物;雄性不育材料可以免除人工去雄和机械去雄对植株的损伤,提高杂交制种的纯度和产量,具有重大商业价值;因此玉米雄性不育的研究在基础理论与应用实践中均具有重要意义。
花药是植物花器官的重要组成部分,与其他花器官相比,被子植物的花药在个体发生、细胞类型、花药形态等方面更为保守 [10]。近几年,拟南芥、水稻、玉米中大量花药发育相关基因被报道,但在解析花药的发育调控机制方面还有很多复杂问题有待解决,且不同物种间花药发育基因功能保守性与特异性还需进一步探索。而在解决这些科学问题的过程中,花药发育的研究方法手段和技术策略显得特别重要,下面分别从玉米花药的形态结构、花药发育时期、花药败育类型、花药发育的细胞学研究方法、组学研究方法、分子遗传学研究方法等方面进行详细阐述。
1 玉米花药的形态结构
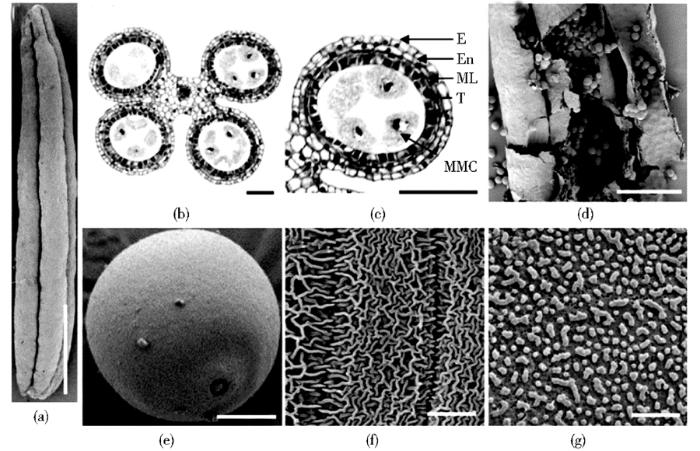
玉米是典型的雌雄同株异花植物,如图1所示,最上面的部位称为雄穗(tassel;图 1b),由数百个成对分布的小穗(spikelet;图1c)组成,每个小穗中包含两个小花(floret),根据发育时期分别被称为上位花和下位花,每个小花由3个花药(anther;图 2a)组成。一般来说,相邻小穗中的花药发育时期高度同步,下位花发育比上位花发育晚约一天 [11,12]。玉米花药由辐射状对称排列的孢子体和配子体两部分组成(图2b),孢子体部分包括连接4个花药小室的连接组织、维管束及花药壁,花药壁由4层体细胞(图 2c)组成,由外向内依次是表皮层(epidermis, E)、内皮层(endothecium, En)、中间层(middle layer, ML)和绒毡层(tapetum, T)。将花药剥开(图2d),内部含有大量的花粉粒(图 2e)。目前,关于绒毡层的相关研究报道较多,受研究材料和技术方法的限制,人们对其他3层细胞的精确功能还缺乏了解,但不可否认,每一层壁细胞的发育均对花药的育性有重要影响。
图1
图1
玉米雄穗和小花形态
Fig.1
Morphology of maize tassel and spikelet
(a) Maize plant at anthesis (b) Tassel of maize (c) Spikelet of maize
图2
图2
玉米花药的形态
Fig.2
Morphology of maize anther
(a) Maize anther (b) Transverse section of maize anther (c) Anther locule (d) Stripped anther (e) Maize pollen granule (f) The outer surface of maize anther (g) The inner surface of maize anther E: Epidermis; En: Endothecium; ML: Middle layer; MMC: Microspore mother cell; T: Tapetum. Bars = 1mm in (a), 30μm in (b,c), 500μm in (d), 20μm (e) and 10μm in (f,g)
1.1 表皮层
1.2 内皮层
1.3 中间层
1.4 绒毡层
绒毡层位于花药壁最内侧,由次生壁细胞内层发育而来,是四层壁细胞中代谢最为活跃的一层。花药发育过程中,绒毡层细胞质浓缩,呈现出腺细胞的特征,内部代谢旺盛,常见细胞中含有双核。减数分裂后可见绒毡层靠近小孢子一侧外面排布有大量的乌氏小体(图 2g)。绒毡层发育缺陷型雄性不育是已报道的植物雄性不育中最多的一类 [19]。孢子母细胞的减数分裂过程离不开绒毡层细胞的支持,绒毡层分泌的葡聚糖可以降解胼胝质,使小孢子得以释放 [20]。减数分裂后绒毡层内进行脂类及其衍生物代谢,产生的物质可作为孢子花粉素合成、加工、组装的原材料,最后通过乌氏小体等运输方式在小孢子外侧积累,形成化学性质极为稳定的花粉粒外壁(图 2e)。
1.5 孢原细胞
孢原细胞(archesporial cell, Ar)位于花药小室中央,起始于L2层细胞的非对称分裂。玉米中起始孢原细胞数量并不确定,经数轮有丝分裂形成小孢子母细胞(microspore mother cell, MMC)后可进入减数分裂[5]。通过EdU(5-乙炔基-2'脱氧尿嘧啶核苷)染色发现,同一花药小室内孢原细胞分裂的节奏并不一致,有些孢原细胞成熟分化为孢子母细胞,而有些孢原细胞会继续分裂;这说明孢原细胞分裂具有随机性,并未受专一调控。进入减数分裂后,同一花药中孢子母细胞染色体行为保持高度一致。
图3
图3
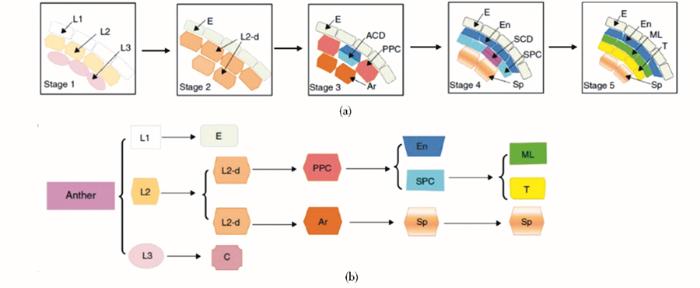
花药发育早期世系假说模式图[34]
Fig.3
Diagram of cell lineages during early anther development
ACD: Asymmetric cell division; Ar: Archesporial cell; C: Connective tissue; E: Epidermis; En: Endothecium; L2-d: L2-derived cell; ML: Middle layer; PPC: Primary parietal cell; SCD: Symmetric cell division; Sp: Sporogenous cell; SPC: Secondary parietal cell; T: Tapetum
2 玉米花药发育时期的划分
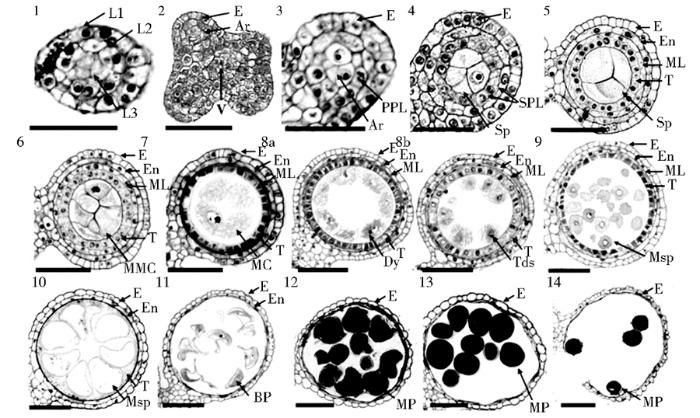
玉米花药的发育起始于雄蕊原基形成,至最后形成成熟的花粉粒,该过程持续约30天,需要孢子体与配子体的协同配合,完成复杂的世代交替过程。根据细胞学形态观察的结果,Warmke和Lee[21]将玉米花药发育过程划分为9个时期,分别为前胼胝质期、中胼胝质期、减数分裂期、四分体期、幼龄小孢子期、单核小孢子期、单核小孢子晚期、幼龄花粉粒期和近成熟花粉粒期,并对各时期的细胞学特征进行描述。Kelliher和Walbot[5]则对减数分裂前玉米花药各层细胞的分化及分裂过程进行详尽观察,并将其划分为8个时期。Zhang等[22,23]结合拟南芥花药发育时期的划分,将禾本科植物水稻花药发育过程细分为14个时期。我们通过半薄切片技术对玉米花药的发育过程进行观察,并与水稻的发育过程进行比对,发现玉米花药横切面的变化与水稻高度一致,因此也将玉米花药发育过程划分为14个时期(图3、图4),各时期细胞学形态特征如下:
(1)时期1(Stage 1):雄蕊原基的起始;花分生组织分化出圆形的雄蕊原基,包含L1、L2、L3三层细胞。
(2)时期2(Stage 2):花药原基分化;雄蕊原基分化形成近长方形的花药原基,此时,L2层细胞在每个角的中心形成孢原细胞。
(3)时期3(Stage 3):孢原细胞经过平周分裂形成原生壁细胞(primary parietal cell, PPL)。
(4)时期4(Stage 4):原生壁细胞再经过一次平周分裂形成两层次生壁细胞(secondary parietal cell, SPL),外层为内皮层;孢原细胞分裂形成造孢细胞(sporogenous cell, Sp)。
(5)时期5(Stage 5):内层次生壁细胞经过平周分裂形成中间层和绒毡层。
(6)时期6(Stage6):造孢细胞分化形成小孢子母细胞,周围被四层花药壁细胞包裹。
(7)时期7(Stage 7):小孢子母细胞与绒毡层接触,减数分裂起始;中间层变成带状结构,并逐渐消失。
(8)时期8a(Stage 8a):减数第一次分裂结束,形成二分体;此时,绒毡层细胞质浓缩,着色加深。
(9)时期8b(Stage 8b):减数第二次分裂结束,形成四分体,小孢子细胞外表面形成初级花粉外壁结构,且被胼胝质紧紧包裹;绒毡层近一步浓缩,且液泡化。
(10)时期9(Stage 9):胼胝质降解,圆形单倍体小孢子被释放,且外表面形成较薄的花粉外壁结构;绒毡层浓缩后在靠近小孢子的内侧表面形成可见的乌氏小体(Ubisch body, Ub)。
(11)时期10(Stage 10):绒毡层进一步降解,产生大量高电子密度的乌氏小体;小孢子中央形成大液泡。
(12)时期11(Stage 11):小孢子经历第一次有丝分裂,产生一个生殖核、一个营养核;绒毡层几乎消失,花药壁内侧可见残存的绒毡层碎片以及乌氏小体。
(13)时期12(Stage 12):生殖核经历第二次有丝分裂,形成成熟的三核花粉粒;绒毡层彻底消失。
(14)时期13(Stage 13):植株表现为开花,花粉囊相连,花药开裂。
(15)时期14(Stage 14):花药散出花粉粒。
3 玉米花药发育的世系假说(linage hypothesis)
花药由4层壁细胞及中央生殖细胞组成(图 2c),各层细胞规则排列且发育受到严格调控,是研究细胞分化的理想模型[33];但被子植物花药发育相关研究多集中于四层壁细胞形态建成之后,关于壁细胞分化过程的报道较少,目前最广为接受的便是世系假说(图3)。该假说强调细胞的世系,认为受到严格调控的平周分裂是花药不同层细胞形态建成的决定因素[35],L2层细胞经过3次平周分裂形成内皮层、中间层、绒毡层以及中央孢原细胞。首先,位于表皮层下的L2层细胞经过第一次平周分裂,形成中央孢原细胞和原生壁细胞。原生壁细胞紧邻表皮层,孢原细胞位于内侧;前者经过短暂的分裂,形成完整包裹孢原细胞的细胞层。该层细胞再经过一次平周分裂,形成内皮层和次生壁细胞;后者经过第三次平周分裂,形成中间层和绒毡层。目前,细胞学观察结果仅能证实世系假说第三次平周分裂,更早期花药发育细胞分化的方向及顺序并未得到证实[36];此外,Kelliher和Walbot[5]通过激光共聚焦显微镜对玉米早期花药发育进行观察,发现孢原细胞并非单一起源,L2层细胞可能会分裂同时产生多个孢原细胞。
图4
图4
玉米花药发育14个时期的细胞学观察
Fig.4
Cytological observation maize anther development at 14 stages
L1, L2, and L3: The three cell layers in stamen primordia; Ar: Archesporial cell; BP: Bicellular pollen; Dy: Dyad cell; E: Epidermis; En: Endothecium; MC: Meiotic cell; ML: Middle layer; MMC: Microspore mother cell; MP: Mature pollen; Msp: Microspore parietal cell; V: Vascular bundle; PPL: Primary parietal layer; SPL: Secondary parietal cell layer; Sp: Sporogenous cell; T: Tapetum; Tds: Tetrads; Bars=50μm
表1 玉米花药发育的细胞学变化过程[10]
Table 1
| 时期 | 形态特征 | 花药长度(μm) | 表达基因 |
|---|---|---|---|
| 1 | 花药原基起始,由L1、L2、L3三层细胞构成 | 30~120 | |
| 2 | L2层细胞分化形成AR细胞 | 120~180 | MSCA1[26] |
| 3 | AR细胞分裂形成PPL | 180~280 | |
| 4 | 细胞分裂继续进行,形成EN和SPC,垂周分裂使花药小室增大 | 280~500 | MAC1[4] |
| 5 | SPL分裂,形成ML和T | 500~700 | |
| 6 | 花药小室增大,AR发育成熟形成MMC | 1 000~1 200 | |
| 7 | MC与绒毡层接触,周围胼胝质降解ML变薄 | 1 200~1 500 | |
| 8a | 减数第一次分裂完成,形成二分体;绒毡层染色质浓缩 | 1 500~2 000 | MS9[27]、MS23[28]、MS32[29] |
| 8b | 减数第二次分裂结束,形成由胼胝质包裹的四分体 | 2 000~2 500 | MS8[30] |
| 9 | 胼胝质降解,小孢子释放 | 2 500~3 000 | MS26[31]、MS45[32]、APV1[25]、IPE1[24] |
| 10 | 小孢子内部形成中央大液泡,与绒毡层接触,绒毡层进一步降解 | 3 000~3 500 | |
| 11 | 小孢子内液泡收缩,细胞核经过一次有丝分裂形成营养核和生殖核 | 3 500~5 000 | |
| 12 | 生殖核在经过一次有丝分裂,形成三核花粉粒;绒毡层彻底降解 | 5 000~5 500 | |
| 13 | 花粉粒继续进行淀粉积累,呈圆球状 | 5 500~6 000 | |
| 14 | 花药开裂,散粉 | 5 500~6 000 |
Note:MSCA1 = Male sterile converted anther1, MAC1 = Multiple archesporial cell1, APV1 = Abnormal pollen vacuolation1, IPE1 = Irregular pollen exine1
4 玉米花药败育类型
此外,已报道的雄性不育可分为三种类型:花药形成受阻(无花药型)、花药结构异常(无花粉型)、花粉粒发育异常(花粉粒败育型)。(1)无花药型突变体:主要表现为花药缺失,雌穗花丝增多,玉米SILKY1和STERILE TASSEL EAR1分别是拟南芥中AP3和PI的同源基因,属于ABC模型中的B家族成员,该类基因突变后会导致雄蕊原基发育受阻,转化为雌蕊原基[38,39]。(2)无花粉型突变体:表现为花药数目减少,且每个花药中花药小室数目减少;未能形成辐射状对称的四层壁细胞,或花药细胞层提前降解;MSCA1基因编码一个谷氧还蛋白,其突变后花药中未能形成孢原细胞,而是分化形成薄壁细胞[26];MAC1基因编码一个小的分泌蛋白,其突变后玉米花药小且孢原细胞数目增多,而体细胞数量减少[4];MS23和MS32基因均编码具有螺旋-环-螺旋结构(basic helix-loop-helix, bHLH)的转录因子,参与调控花药壁分化过程,它们的突变会导致绒毡层细胞发育异常[28-29, 40];MS8基因编码一个 -1,3-半乳糖基转移酶,在减数分裂时期的花药中大量表达,ms8突变体表现为花药伸长受阻,绒毡层细胞数目减少,体积增大,胼胝质增多,孢子细胞完成减数第一次分裂后降解 [20,30];此类突变体败育时期较早,孢子母细胞未能进入减数分裂或者未能完成减数分裂过程。(3)花粉粒发育异常型雄性不育突变体:也称为功能缺失型雄性不育突变体。该类突变体组织发生正常,可形成辐射状对称的四层壁细胞和中央的孢子母细胞,且孢子母细胞能顺利完成减数分裂,但随后小孢子发育出现异常。小孢子外壁由孢子花粉素堆积形成,其主要成分为脂类及其衍生物,在绒毡层合成,然后运输至小孢子外侧。IPE1[24]、APV1[25]、MS26[31]、MS45[32]等基因编码脂肪酸及其衍生物代谢相关的氧化还原酶,参与花粉外壁主要成分孢子花粉素的合成,该类基因突变会导致小孢子外壁发育受阻。此外,ipe1、apv1和ms26突变体还表现为花药外壁光滑、角质层消失[24,25],说明花药角质层与孢子花粉素共用绒毡层中的脂类代谢途径。
5 玉米花药发育的细胞学研究方法
对表型的精细观察是花药发育研究的前提,此部分对组织、细胞、亚细胞等不同层面的细胞学研究方法进行了总结。
5.1 扫描电镜(scanning electron microscope, SEM)观察
SEM可用于观察花药及花粉粒表面形态,主要是用于判断花药表面角质层、花药内壁乌氏小体、花粉粒及其外壁的发育。具体操作过程可分为材料固定、梯度脱水、干燥、镀金和观察分析5个步骤。取野生型和突变体不同发育时期花药,浸于FAA(formalin-aceto-alcohol)固定液中,固定液的体积不少于所取研究材料体积的20倍;对于已变黄的花药,可用解剖针在花药壁穿孔以提高固定液的渗透效果,或者反复抽真空至花药沉入固定液底部;室温固定2h后,将材料置于4℃保存,或依次置于50%、60%、70%、80%、90%、100%的乙醇中进行脱水,每个梯度保持15min;材料可置于70%的乙醇中过夜或保存。脱水后的样品进行二氧化碳临界点干燥,然后镀金即可进行观察。
5.2 树脂半薄切片观察
树脂切片技术在花药发育相关研究中具有重要用途,可判断花药发育时期,突变体组织层面表型异常,其观察分析结果可有效地指导后续实验材料的选取。用于树脂切片的花药固定过程与SEM材料固定一致,随后利用梯度乙醇(50%、70%、90%、100%)进行脱水,每步15~30min。脱水过程中花药可置于70%乙醇中长期保存;为便于后期包埋,可在90%乙醇中加入0.1%的伊红对材料进行染色;为保证脱水彻底,材料须在无水乙醇中脱水2~3次。随后进行树脂替换,将花药依次置于乙醇与Spurr树脂体积比为3∶1、1∶1、1∶3液体中2~4h,最后置于纯树脂中过夜。树脂置换完成后,将花药置于模具中,加入200μl Spurr树脂,置于烘箱中,70℃聚合过夜。随后进行修块,然后可利用德国莱卡切片机进行切片,切片厚度为2μm;用镊子夹取切好的片子,置于载玻片中央的无菌水中,42℃条件下展片过夜。将固定有样品的载玻片浸入0.1%的甲苯胺蓝染液中,染色1min,然后用去离子水冲洗,再置于展片台上,烘干后即可用于显微观察;也可以封片后长期保存。
5.3 透射电镜(transmission electron microscope, TEM)观察
为观察花药亚细胞层面的结构或突变体更为精细的结构变异,需根据半薄切片的结果,选择合适时期的花药进行TEM观察。具体操作过程如下:将玉米花药浸入预冷的戊二醛固定液中,反复抽真空,至样品完全浸入固定液,4℃条件下固定2h,更换固定液,置于室温中过夜,利用0.1mol/L的PBS漂洗4次,每次30min;去除PBS后,加入2%的锇酸固定液,通风厨中室温固定4h;去除锇酸固定液后,0.1mol/L PBS漂洗4次,每次20min;后续乙醇梯度脱水、树脂置换、包埋的过程同半薄树脂切片制作过程一致;修块后,利用超薄切片机进行切片,片子厚度为70nm,切片所用玻璃刀或钻石刀需安装水槽,进行展片,随后使用睫毛笔将片子转移至镀有聚醋酸甲基乙烯脂膜的铜网上,并将铜网置于干燥环境中保存;将铜网粘有片子的一面浸入2%醋酸双氧铀染液中,染色15min,随后使用双蒸水漂洗3次,每次1min;再将铜网置于柠檬酸铅染液中,染色15min,随后再使用双蒸水漂洗3次,每次1min,使用吸水纸吸去多余的水分,干燥环境中保存或干燥后利用透射电子显微镜进行观察分析。
5.4 孢子细胞染色体DAPI(4',6-diamidino-2-phenylindole)染色
孢子母细胞经过一次减数分裂形成小孢子,随后小孢子经过两次有丝分裂形成包含一个营养核、两个生殖核的成熟花粉粒。直接参与减数分裂或有丝分裂的基因发生突变都可能会导致雄性不育之外的其他表型,本文不做讨论。花药发育过程中小孢子与绒毡层具有紧密联系,某些基因虽不直接参与减数分裂或有丝分裂,但基因突变后也会影响减数分裂的进程,在对该类突变体进行细胞学研究时,需对其染色体行为进行DAPI染色观察。具体操作过程为:将减数分裂时期的花药浸没于新配制的卡诺固定液(无水乙醇∶冰醋酸=3∶1)中,-20℃条件下固定24h;将花药置于载玻片上,加10μl醋酸洋红染液,破碎并夹出花药壁,盖上盖玻片后置于光学显微镜下观察;选择处于减数分裂时期的样品,做好标记,置于液氮中速冻,去除盖玻片,-20℃保存或加5μl DAPI染液,染色10min后置于荧光显微镜下观察。若要对减数分裂后小孢子内细胞核数量进行检测,则需取不同时期的花药浸没于FAA固定液中,室温固定12h;首先根据花药长度初步判断花药发育时期,然后选择合适的花药置于载玻片上,滴加5μl DAPI染液,用镊子轻轻挤出小孢子后染色10min,利用荧光显微镜进行观察分析。
6 玉米花药发育的组学研究方法
随着科技的进步,利用多种组学手段和高通量测序技术对玉米不育材料进行深入研究,为人们解析复杂的雄性不育调控机制提供了便利条件,本文同样也对转录组、蛋白质组、代谢组技术在花药发育研究中的应用进行了总结。
6.1 玉米花药脂类代谢产物含量测定分析
角质层包含角质层蜡和角质两部分。角质层蜡是由长链脂肪族化合物组成的混合物;角质则主要由脂肪酸衍生物多聚体构成,且被角质层蜡包裹[41,42]。花粉外壁包裹在花粉粒最外层,是植物中最为复杂的细胞壁结构,由理化性质极为稳定的孢子花粉素构成,由于其超稳定的理化性质,孢子花粉素的精确组成成分还未知;近几年,随着新的物理、化学技术的出现,如磁共振、硫代氨基甲酸酯处理,人们对孢子花粉素的组成有了进一步了解。越来越多的证据表明,孢子花粉素是由多种脂肪族单体物质构成,包括长链脂肪酸及其多羟基化衍生物、酚醛树脂类复合物等。这也就意味着脂肪酸代谢在角质层和花粉外壁的合成过程中发挥重要作用 [43],因此对花药中蜡质、角质及游离脂肪酸的含量测定有助于解析参与该过程相关基因的功能。
在脂类物质含量测定之前,首先建立花药鲜重与表面积之间的线性关系 [44],将花药看做圆柱体,称取不同质量的花药,进行扫描,然后统计各个花药的长与宽,分别作为圆柱体的高与底面直径,最后计算不同质量花药对应总表面积,进行线性化作图。脂类物质含量测定的具体过程如下[45]:(1)分别称取野生型和突变体各200mg鲜重花药置于玻璃瓶中,加入3ml氯仿浸提1min,抽提液中加入10μg十七烷酸和30μg正二十二烷醇作为内标物,转移至新的玻璃瓶中,氮气吹干后,加入100μl MSTFA(N-methyl-N-trimethylsilyl-trifluoroacetamide),50℃条件下衍生60分钟,使提取物中的自由羟基和羧基转化为相应的三甲基硅醚/酯,离心后取上清,进行GC-MS分析。(2)将上述花药重新浸入3ml氯仿-甲醇溶液(1∶1, V∶V)中,50℃条件下孵育30min,然后室温摇晃72h,提取液于氮气中吹干待用;将脱脂后的花药冷冻干燥,用以分析角质单体。(3)脂肪酸提取物和脱脂花药中同时加入1ml 1个当量的盐酸甲醇溶液,并加入10μg十七烷酸作为内标,80℃条件下孵育2h,再加入2ml饱和氯化钠,混匀后加入1ml正丁烷进行抽提,重复三次;合并抽提液,氮气吹干后,真空干燥4h,残渣中加入100μl MSTFA,50℃条件下衍生60min,离心后取上清,进行GC-MS分析。
6.2 玉米花药转录组和蛋白质组样品选取与组学分析
随着测序技术的发展及成本的降低,转录组和蛋白质组分析成为较普遍的信息挖掘方式;样品的选取对于后续信息分析具有重大影响。虽现已证明花药长度与发育时期具有较强的线性关系 [5],但为提高生物重复之间的一致性,建议基于树脂半薄切片分析结果选取适当时期的样品,以准确判断两个待比较样品的发育时期。
目前,已有多篇文章对玉米花药不同发育时期的转录组及蛋白质组变化进行报道,且主要集中在减数分裂前及减数分裂等花药发育前期[46,47,48]。Ma等[49]对花药起始后小穗、有丝分裂增殖期花药、减数分裂前花药及减数第一次分裂前期花药进行转录本芯片分析,发现减数分裂前期花药转录本复杂性最高,MADS转录因子及富含亮氨酸的受体激酶在花药发育过程中可能发挥重要功能。对上述时期及单核期、双核期、成熟花粉粒转录本分析表明,成熟期花粉粒中转录本最少,减数分裂期转录本复杂性最低;参与花药发育的基因种类繁多,部分基因的表达具有时期特异性,部分基因在花药发育大多数时期均有表达[3]。Zhang等[50]将减数分裂前花药进一步划分为5个时期,并结合纤维切割技术对花药不同层壁细胞转录本进行检测;同时利用质谱技术检测相同发育时期蛋白质组,结果显示花药发育早期蛋白质组与减数分裂期蛋白质组之间存在较大差异,且同时期转录组与蛋白质组一致性较差。利用转录组及蛋白质组学手段研究玉米花药不同发育时期及雄性不育突变体与野生型之间表达基因的差异将为解析玉米花药发育的调控机制提供便利途径。
6.3 玉米花药代谢组学分析
多种初级及次级代谢产物参与玉米花药发育过程。例如,角质层蜡由碳链长度为22~48的长链脂肪族化合物组成。这些化合物的合成依赖于内质网中的延伸复合体[54]。该复合体产生的长链酯酰辅酶A可以经过醇类产生途径,被加工成奇数碳的初级醇和烷脂;或者经过烷烃合成途径,被加工成醛、偶数链烷烃、次级醇和酮类等物质。角质多聚体则主要由内部酯化的羟基脂肪酸构成[55],也包含一些丙三醇、二羧酸和酚醛树脂化合物等物质。孢子花粉素是花粉外壁的主要组成成分,由多种脂肪族单体物质构成,包括长链脂肪酸及其多羟基化衍生物、酚醛树脂类复合物等[56]。虽至今尚无关于玉米花药发育的代谢组报道,但直接测量突变体代谢组或者特定代谢组分的变化以解析相关功能基因已在拟南芥中得到应用。拟南芥中苯丙氨酸代谢途径中的苯丙氨酸脱氨酶(phenylalanine ammonia-lyase, PAL)由4个基因编码,代谢组分析发现pal1pal2双突变体中缺乏3种黄酮醇苷类物质,而苯丙氨酸过量积累,同时其他非芳香族类氨基酸代谢均受到影响,且木质素含量降低[57]。对花药壁及花粉壁异常突变体代谢组的研究将为解析目的基因的功能提供新的视角。
6.4 玉米花药发育的多组学联合分析
随着技术的发展,基于多组学的全转录组测序分析已成为研究趋势。除mRNA外,全转录组分析还包括对miRNA(小RNA)、LncRNA(长非编码RNA)、circRNA(环状RNA)的测序分析。PhasiRNA(phased small-interfering RNA)是小RNA的一种,有21nt和24nt两类,现有研究表明21nt phasiRNA在亚表皮层富集,24nt phasiRNA在绒毡层及孢子细胞中富集;且phasiRNA在不同的花药壁发育异常突变体中发生不同的变化,该研究表明phasiRNA在玉米花药早期发育过程中发挥重要功能[58]。水稻农垦58S为光温敏不育突变体,不育特性由pms1和pms3两个位点控制;近期研究表明PMS1为不完全显性基因,编码一个LncRNA,PMS1能够被microRNA2118识别并介导剪切,形成一串21nt的小RNA。农垦58S与可育品种在pms1区间存在一个突变的碱基,位于剪切位点下游的24bp,这一突变导致农垦58S在长日照下能产生更多的phasiRNA,从而造成雄性不育[59]。由于miRNA及LncRNA主要通过介导目的基因转录本沉默来发挥作用,因此对miRNA、LncRNA靶基因及mRNA差异基因进行联合分析,更有利于解析花药发育的调控网络。CircRNA是最近才被发现的一类RNA,该类RNA 5'端和3'端共价结合形成闭合环状结构,不易被降解,可在细胞内长期行驶转录调控功能;虽然该类RNA在玉米花药发育过程中的功能未见诸报道,但其普遍参与动植物生长发育及对环境胁迫的响应;对该类RNA的研究必将丰富对玉米花药发育调控的认知。
此外,随着分析方法的进步,利用转录组、蛋白质组及代谢组进行联合分析,构建基因、蛋白质与代谢物的互作网络,发掘核心代谢通路,将为解析玉米花药发育调控网络提供更为宏观的视野。
7 玉米花药发育遗传学与分子生物学研究方法
7.1 玉米核不育基因的遗传学分析与图位克隆
玉米雄性不育是田间常见的突变类型。对此类材料进行遗传学分析,需首先利用其他自交系给不育材料授粉,若F1全部可育,则为隐性突变;若F1出现可育、不育分离,该突变可能为显性突变;上述两种突变均属于孢子体类型。若F1植株产生的花粉粒均为一半可育一半不育,则为配子体突变。目前,较为常见的突变类型为孢子体隐性核不育,对于该类型突变体,可利用F1自交构建F2群体,或者给突变体授粉构建BC1F1群体,并对群体植株的育性分离比进行统计。若F2群体中可育单株与不育单株的分离比符合3∶1,或者BC1F1群体中可育单株与不育单株分离比符合1∶1,则为隐性单基因突变体。
遗传学分析完成后,可利用构建的分离群体对单基因突变体进行图位克隆,或利用混合组分分析(bulk segregation analysis,BSA)结合全基因组重测序的方法,筛选目的基因[60]。精细定位完成后可得到候选基因,随后通过转基因或者基因敲除的方式对候选基因进行功能验证。
7.2 基因表达分析
玉米花药发育是一个复杂的生物学过程,至少75%的玉米基因组已注释的基因在玉米花药发育过程中表达[50]。但同时各个基因的表达受到严格调控,因此对于已知基因的时空特异性表达分析有助于解析基因的表达部位与潜在的功能。qRT-PCR可以检测基因特异表达的器官,结合半薄切片分析结果,则可以对基因表达所处花药发育时期进行精确判断。此外,基因表达在组织层面也具有特异性,可利用原位杂交实验检测基因表达的组织特异性,同时与qRT-PCR结果相互验证。
在亚细胞层面,蛋白质需进入不同的细胞器发挥功能;因此对已知蛋白质的亚细胞定位分析,有助于解析其发挥作用的方式。目前,常用的亚细胞定位方式是将目的蛋白与荧光蛋白融合转化原生质体、洋葱表皮细胞或烟草叶片进行瞬时表达,通过检测目的蛋白荧光信号与特定细胞器染料荧光信号的共定位情况,判断该蛋白质的亚细胞定位。随着超薄切片技术的进步,将TEM与蛋白质免疫印迹相结合的免疫胶体金技术(immune colloidal gold technique)提供更为直观的检测蛋白定位的方法。
7.3 基因调控作用机理研究
不同基因以调控网络的形式参与花药的发育过程,因此解析不同基因间相互关系可以帮助我们更宏观地了解玉米发育的调控过程。不同的蛋白质通常以复合体的形式发挥作用,酵母双杂技术(yeast two hybridization)可筛选与目的蛋白相互作用的蛋白质,用以解析蛋白质发挥作用的具体方式。基因的时空特异性表达受上游转录因子的调控,酵母单杂交技术则可以筛选调控基因表达的反式作用因子。染色体免疫沉淀技术(chromosome immunoprecipitation)与二代测序(next generation sequencing)技术相结合的ChIP-seq技术则可以筛选已知转录因子潜在的调控基因。上述研究方法与各种组学研究方法及基因编辑技术相结合,可有效解析基因调控代谢网络。
8 讨论
玉米花药发育的研究在基础理论和应用实践中均具有重要的作用。随着技术的进步,在模式植物拟南芥和水稻中,大量参与花药发育的基因被克隆报道,其调控网络正在初步建立[61,62,63,64]。被子植物花药形态的保守性预示着不同物种间花药发育调控的保守性,CRISPR/Cas9基因组编辑技术的成熟以及在玉米中的成功运用[65],为我们研究同源基因在玉米花药发育过程中的功能提供便捷途径。此外,随着本实验室玉米多控不育(mutli-control sterility, MCS)技术和美国先锋公司SPT(seed production technology)技术的报道,玉米隐性核不育突变体重新得到人们的重视,IPE1 [24]、APV1[25]、MS2[66]、MS7[67]等基因最近相继被报道;这有助于人们更好地理解玉米花药发育的调控机制并指导其在生产中的应用。
参考文献
Plant meiosis: the means to 1N
Meiosis is pivotal in the life history of plants. In addition to providing an opportunity for genetic reassortment, it marks the transition from diploid sporophyte to haploid gametophyte. Recent molecular data suggest that, like animals, plants possess a common set of genes (also conserved in eukaryotic microorganisms) responsible for meiotic recombination and chromosome segregation. However, unlike animals, plant meiocytes do not differentiate from a pool of primordial germ cells, but rather arise de novo from a germline formed from sub-epidermal cells in the anthers and ovules. Mutants defective in the specification of these reproductive cell lines and disrupted in different aspects of the meiotic process are beginning to reveal many features unique to plant meiosis.
Molecular genetic analyses of microsporogenesis and microgametogenesis in flowering plants
Male reproductive development: gene expression profiling of maize anther and pollen ontogeny
During flowering, central anther cells switch from mitosis to meiosis, ultimately forming pollen containing haploid sperm. Four rings of surrounding somatic cells differentiate to support first meiosis and later pollen dispersal. Synchronous development of many anthers per tassel and within each anther facilitates dissection of carefully staged maize anthers for transcriptome profiling. Global gene expression profiles of 7 stages representing 29 days of anther development are analyzed using a 44 K oligonucleotide array querying approximately 80% of maize protein-coding genes. Mature haploid pollen containing just two cell types expresses 10,000 transcripts. Anthers contain 5 major cell types and express >24,000 transcript types: each anther stage expresses approximately 10,000 constitutive and approximately 10,000 or more transcripts restricted to one or a few stages. The lowest complexity is present during meiosis. Large suites of stage-specific and co-expressed genes are identified through Gene Ontology and clustering analyses as functional classes for pre-meiotic, meiotic, and post-meiotic anther development. MADS box and zinc finger transcription factors with constitutive and stage-limited expression are identified. We propose that the extensive gene expression of anther cells and pollen represents the key test of maize genome fitness, permitting strong selection against deleterious alleles in diploid anthers and haploid pollen. Because flowering plants show a substantial bias for male-sterile compared to female-sterile mutations, we propose that this fitness test is general. Because both somatic and germinal cells are transcriptionally quiescent during meiosis, we hypothesize that successful completion of meiosis is required to trigger maturation of anther somatic cells.
Maize multiple archesporial cells 1 (mac1), an ortholog of rice TDL1A, modulates cell proliferation and identity in early anther development
Abstract To ensure fertility, complex somatic and germinal cell proliferation and differentiation programs must be executed in flowers. Loss-of-function of the maize multiple archesporial cells 1 (mac1) gene increases the meiotically competent population and ablates specification of somatic wall layers in anthers. We report the cloning of mac1, which is the ortholog of rice TDL1A. Contrary to prior studies in rice and Arabidopsis in which mac1-like genes were inferred to act late to suppress trans-differentiation of somatic tapetal cells into meiocytes, we find that mac1 anthers contain excess archesporial (AR) cells that proliferate at least twofold more rapidly than normal prior to tapetal specification, suggesting that MAC1 regulates cell proliferation. mac1 transcript is abundant in immature anthers and roots. By immunolocalization, MAC1 protein accumulates preferentially in AR cells with a declining radial gradient that could result from diffusion. By transient expression in onion epidermis, we demonstrate experimentally that MAC1 is secreted, confirming that the predicted signal peptide domain in MAC1 leads to secretion. Insights from cytology and double-mutant studies with ameiotic1 and absence of first division1 mutants confirm that MAC1 does not affect meiotic cell fate; it also operates independently of an epidermal, Ocl4-dependent pathway that regulates proliferation of subepidermal cells. MAC1 both suppresses excess AR proliferation and is responsible for triggering periclinal division of subepidermal cells. We discuss how MAC1 can coordinate the temporal and spatial pattern of cell proliferation in maize anthers.
Emergence and patterning of the five cell types of the Zea mays anther locule
One fundamental difference between plants and animals is the existence of a germ-line in animals and its absence in plants. In flowering plants, the sexual organs (stamens and carpels) are composed almost entirely of somatic cells, a small subset of which switch to meiosis; however, the mechanism of meiotic cell fate acquisition is a long-standing botanical mystery. In the maize (Zea mays) anther microsporangium, the somatic tissues consist of four concentric cell layers that surround and support reproductive cells as they progress through meiosis and pollen maturation. Male sterility, defined as the absence of viable pollen, is a common phenotype in flowering plants, and many male sterile mutants have defects in somatic and reproductive cell fate acquisition. However, without a robust model of anther cell fate acquisition based on careful observation of wild-type anther ontogeny, interpretation of cell fate mutants is limited. To address this, the pattern of cell proliferation, expansion, and differentiation was tracked in three dimensions over 30 days of wild-type (W23) anther development, using anthers stained with propidium iodide (PI) and/or 5-ethynyl-2'-deoxyuridine (EdU) (S-phase label) and imaged by confocal microscopy. The pervading lineage model of anther development claims that new cell layers are generated by coordinated, oriented cell divisions in transient precursor cell types. In reconstructing anther cell division patterns, however, we can only confirm this for the origin of the middle layer (ml) and tapetum, while young anther development appears more complex. We find that each anther cell type undergoes a burst of cell division after specification with a characteristic pattern of both cell expansion and division. Comparisons between two inbreds lines and between ab- and adaxial anther florets indicated near identity: anther development is highly canalized and synchronized. Three classical models of plant organ development are tested and ruled out; however, local clustering of developmental events was identified for several processes, including the first evidence for a direct relationship between the development of ml and tapetal cells. We speculate that small groups of ml and tapetum cells function as a developmental unit dedicated to the development of a single pollen grain.
Anther development: basic principles and practical applications
Male reproductive processes in flowering plants take place in the stamen (Esau, 1977). This sporophytic organ system contains diploid cells that undergo meiosis and produce haploid male spores, or microspores. Microspores divide mitotically
Stamen structure and function
玉米隐性核雄性不育基因研究进展及其育种应用途径分析
玉米隐性核雄性不育材料是玉米生产和育种中广泛存在的一种特异的种质资源,对玉米杂交育种和杂交种生产都具有极其重要的意义,但是由于这类不育材料缺乏有效的保持和繁殖技术体系,长期以来一直未能在玉米遗传育种和杂交制种过程中得到充分利用。本文拟从玉米隐性核不育基因的研究历史、现状及未来可能的育种应用途径等方面逐一概述,并整合现代生物技术手段和常规杂交育种策略,以期为解决玉米隐性核不育基因的有效育种利用、建立高效的玉米杂交育种体系和降低杂交种制种成本等关键科学和技术问题提供设计思路和理论依据。
Research progress on maize recessive genic male sterility gene and its utilization strategies in maize breeding program
玉米隐性核雄性不育材料是玉米生产和育种中广泛存在的一种特异的种质资源,对玉米杂交育种和杂交种生产都具有极其重要的意义,但是由于这类不育材料缺乏有效的保持和繁殖技术体系,长期以来一直未能在玉米遗传育种和杂交制种过程中得到充分利用。本文拟从玉米隐性核不育基因的研究历史、现状及未来可能的育种应用途径等方面逐一概述,并整合现代生物技术手段和常规杂交育种策略,以期为解决玉米隐性核不育基因的有效育种利用、建立高效的玉米杂交育种体系和降低杂交种制种成本等关键科学和技术问题提供设计思路和理论依据。
Unresolved issues in pre-meiotic anther development
Compared to the diversity of other floral organs, the steps in anther ontogeny, final cell types, and overall organ shape are remarkably conserved among Angiosperms. Defects in pre-meiotic anthers that alter cellular composition or function typically result in male-sterility. Given the ease of identifying male-sterile mutants, dozens of genes with key roles in early anther development have been identified and cloned in model species, ordered by time of action and spatiotemporal expression, and used to propose explanatory models for critical steps in cell fate specification. Despite rapid progress, fundamental issues in anther development remain unresolved, and it is unclear if insights from one species can be applied to others. Here we construct a comparison of Arabidopsis, rice, and maize immature anthers to pinpoint distinctions in developmental pace. We analyze the mechanisms by which archesporial (pre-meiotic) cells are specified distinct from the soma, discuss what constitutes meiotic preparation, and review what is known about the secondary parietal layer and its terminal periclinal division that generates the tapetal and middle layers. Finally, roles for small RNAs are examined, focusing on the grass-specific phasiRNAs.
Petal and stamen development
Expression of MADS box genes ZMM8 and ZMM14 during inflorescence development of Zea mays discriminates between the upper and the lower floret of each spikelet
Most floral meristem and organ identity genes of dicotyledonous plants belong to the MADS box gene family. Since they are generally transcribed in those tissues and organs whose identity they determine, they are excellent markers for developmental processes. Here we report the cDNA cloning of a pair of MADS box genes, ZMM8 and ZMM14 , from the monocotyledonous plant maize. Maize inflorescences are composed of spikelets which contain two florets, an upper and a lower one. Although upper and lower florets develop in a very similar way in male inflorescences, ZMM8 and ZMM14 expression was found in all organs of upper florets, but no transcripts were detected in lower florets. In contrast, two other MADS box genes were found to be expressed in lower florets in the same way as in upper florets. Our observations suggest that during spikelet development ZMM8 and ZMM14 work as selector genes which are involved in distinguishing the upper from the lower floret. Alternatively, these genes may be involved in conferring determinacy to the spikelet or upper floret meristem. Our data suggest that in the phylogenetic lineage that led to maize an ancient type of MADS box gene has been recruited during evolution for the establishment of novel positional information not found within the simple inflorescences of dicotyledonous plants such as Arabidopsis .
Perception of free cutin monomers by plant cells
Enzymatic degradation of plant cuticles by fungal pathogens results in the release of free cutin monomers. The hypothesis that free cutin monomers are recognized by plant cells as endogenous stress-related signals was tested in a model system consisting of cultured potato cells. Addition of cutin monomers in the micromolar range induced a transient alkalinization of the culture medium, similar to that observed with chitin or chitotetraose that served as positive control. The cutin monomers tested varied considerably in their potential to induce alkalinization, the most and least active compounds being cis -9,10-epoxy-18-hydroxystearic acid and palmitic acid, respectively. n ,16-dihydroxypalmitic acid ( n = 8, 9 or 10) was found to be the major component of potato leaf cuticle and was among the most active cutin monomers. 9,10-Dihydroxystearic acid, an analogue of the cutin monomer threo -9,10,18-trihydroxystearic acid, exhibited biological activity in a stereoselective manner, only the naturally occurring threo -stereoisomer inducing a rapid and strong alkalinization response. Alkalinization of the culture medium was inhibited by addition of the protein-kinase inhibitor K-252a, and the onset of alkalinization was paralleled by changes in phosphorylation of specific proteins. The active cutin monomers also stimulated the production of the plant stress hormone ethylene and activated defence-related genes at the mRNA level. The data provide evidence for a role of enzymatic breakdown products of plant cuticles as a new class of endogenous signal molecules.
Cutin monomers and surface wax constituents elicit H2O2 in conditioned cucumber hypocotyl segments and enhance the activity of other H2O2 elicitors
The ANTHER INDEHISCENCE1 gene encoding a single MYB domain protein is involved in anther development in rice
Using a two-element iAc/Ds transposon-tagging system, we identified a rice (Oryza sativa L. cv Nipponbare) recessive mutant, anther indehiscence1 (aid1), showing partial to complete spikelet sterility. Spikelets of the aid1 mutant could be classified into three types based on the viability of pollen grains and the extent of anther dehiscence. Type 1 spikelets (approximately 25%) were sterile due to a failure in accumulation of starch in pollen grains. Type 2 spikelets (approximately 55%) had viable pollen grains, but anthers failed to dehisce and/or synchronize with anthesis due to failure in septum degradation and stomium breakage, resulting in sterility. type 3 spikelets (approximately 20%) had normal fertility. In addition, aid1 mutant plants had fewer tillers and flowered 10 to 15 d later than the wild type. The Ds insertion responsible for the aid1 mutation was mapped within the coding region of the AID1 gene on chromosome 6, which is predicted to encode a novel protein of 426 amino acids with a single MYB domain. The MYB domain of AID1 is closely related to that of the telomere-binding proteins of human, mouse, and Arabidopsis, and of single MYB domain transcriptional regulators in plants such as PcMYB1 and ZmIBP1. AID1 was expressed in both the leaves and panicles of wild-type plants, but not in mutant plants.
The mac1 mutation alters the developmental fate of the hypodermal cells and their cellular progeny in the maize anther
Abstract In angiosperm ovules and anthers, the hypodermal cell layer provides the progenitors of meiocytes. We have previously reported that the multiple archesporial cells1 (mac1) mutation identifies a gene that plays an important role in the switch of the hypodermal cells from the vegetative pathway to the meiotic (sporogenous) pathway in maize ovules. Here we report that the mac1 mutation alters the developmental fate of the hypodermal cells of the maize anther. In a normal anther a hypodermal cell divides periclinally with the inner cell giving rise to the sporogenous archesporial cells while the outer cell, together with adjacent cells, forms the primary parietal layer. The cells of the parietal layer then undergo two cycles of periclinal divisions to give rise to three wall layers. In mac1 anthers the primary parietal layer usually fails to divide periclinally so that the three wall layers do not form, while the archesporial cells divide excessively and most fail to form microsporocytes. The centrally located mutant microsporocytes are abnormal in appearance and in callose distribution and they fail to proceed through meiosis. These failures in development and function appear to reflect the failure of mac1 gene function in the hypodermal cells and their cellular progeny.
Overexpression of TAPETUM DETERMINANT1 alters the cell fates in the Arabidopsis carpel and tapetum via genetic interaction with excess microsporocytes1/extra sporogenous cells
Previously, we reported that the TAPETUM DETERMINANT1 (TPD1) gene is required for specialization of tapetal cells in the Arabidopsis (Arabidopsis thaliana) anther. The tpd1 mutant is phenotypically identical to the excess microsporocytes1 (ems1)/extra sporogenous cells (exs) mutant. The TPD1 and EMS1/EXS genes may function in the same developmental pathway in the Arabidopsis anther. Here, we further report that overexpression of TPD1 alters the cell fates in the Arabidopsis carpel and tapetum. When TPD1 was expressed ectopically in the wild-type Arabidopsis carpel, the number of cells in the carpel increased significantly, showing that the ectopic expression of TPD1 protein could activate the cell division in the carpel. Furthermore, the genetic analysis showed that the activation of cell division in the transgenic carpel by TPD1 was dependent on EMS1/EXS, as it did not happen in the ems1/exs mutant. This result further suggests that TPD1 regulates cell fates in coordination with EMS1/EXS. Moreover, overexpression of TPD1 in tapetal cells also delayed the degeneration of tapetum. The TPD1 may function not only in the specialization of tapetal cells but also in the maintenance of tapetal cell fate.
Receptor-like protein kinase 2 (RPK 2) is a novel factor controlling anther development in Arabidopsis thaliana
Summary Receptor-like kinases (RLK) comprise a large gene family within the Arabidopsis genome and play important roles in plant growth and development as well as in hormone and stress responses. Here we report that a leucine-rich repeat receptor-like kinase (LRR-RLK), RECEPTOR-LIKE PROTEIN KINASE2 (RPK2), is a key regulator of anther development in Arabidopsis. Two RPK2 T-DNA insertional mutants ( rpk2-1 and rpk2-2 ) displayed enhanced shoot growth and male sterility due to defects in anther dehiscence and pollen maturation. The rpk2 anthers only developed three cell layers surrounding the male gametophyte: the middle layer was not differentiated from inner secondary parietal cells. Pollen mother cells in rpk2 anthers could undergo meiosis, but subsequent differentiation of microspores was inhibited by tapetum hypertrophy, with most resulting pollen grains exhibiting highly aggregated morphologies. The presence of tetrads and microspores in individual anthers was observed during microspore formation, indicating that the developmental homeostasis of rpk2 anther locules was disrupted. Anther locules were finally crushed without stomium breakage, a phenomenon that was possibly caused by inadequate thickening and lignification of the endothecium. Microarray analyses revealed that many genes encoding metabolic enzymes, including those involved in cell wall metabolism and lignin biosynthesis, were downregulated throughout anther development in rpk2 mutants. RPK2 mRNA was abundant in the tapetum of wild-type anthers during microspore maturation. These results suggest that RPK2 controls tapetal cell fate by triggering subsequent tapetum degradation, and that mutating RPK2 impairs normal pollen maturation and anther dehiscence due to disruption of key metabolic pathways.
Floret-specific differences in gene expression and support for the hypothesis that tapetal degeneration of Zea mays L. occurs via programmed cell death
The maize (Zea mays) spikelet consists of two florets, each of which contains three developmentally synchronized anthers. Morphologically, the anthers in the upper and lower florets proceed through apparently similar developmental programs. To test for global differences in gene expression and to identify genes that are coordinately regulated during maize anther development, RNA samples isolated from upper and lower floret anthers at six developmental stages were hybridized to eDNA rnicroarrays. Approximately 9% of the tested genes exhibited statistically significant differences in expression between anthers in the upper and lower florets. This finding indicates that several basic biological processes are differentially regulated between upper and lower floret anthers, including metabolism, protein synthesis and signal transduction. Genes that are coordinately regulated across anther development were identified v/a cluster analysis.Analysis of these results identified stage-specific, early in development, late in development and bi-phasic expression profiles. Quantitative RT-PCR analysis revealed that four genes whose homologs in other plant species are involved in programmed cell death are up-regulated just prior to the time the tapetum begins to visibly degenerate (i.e., the mid-microspore stage). This finding supports the hypothesis that developmentally normal tapetal degeneration occurs via programmed cell death.
The male sterile 8 mutation of maize disrupts the temporal progression of the transcriptome and results in the mis-regulation of metabolic functions
Abstract Maize anther ontogeny is complex, with the expression of more than 30,000 genes over 4 days of cell proliferation, cell fate acquisition and the start of meiosis. Although many male-sterile mutants disrupt these key steps, few have been investigated in detail. The terminal phenotypes of Zea mays (maize) male sterile 8 (ms8) are small anthers exhibiting meiotic failure. Here, we document much earlier defects: ms8 epidermal cells are normal in number but fail to elongate, and there are fewer, larger tapetal cells that retain, rather than secrete, their contents. ms8 meiocytes separate early, have extra space between them, occupied by excess callose, and the meiotic dyads abort. Thousands of transcriptome changes occur in ms8, including ectopic activation of genes not expressed in fertile siblings, failure to express some genes, differential expression compared with fertile siblings and about 40% of the differentially expressed transcripts appear precociously. There is a high correlation between mRNA accumulation assessed by microarray hybridization and quantitative real-time reverse transcriptase polymerase chain reaction. Sixty-three differentially expressed proteins were identified after two-dimensional gel electrophoresis followed by liquid chromatography tandem mass spectroscopy, including those involved in metabolism, plasmodesmatal remodeling and cell division. The majority of these were not identified by differential RNA expression, demonstrating the importance of proteomics in defining developmental mutants.
Mitochondrial degeneration in Texas cytoplasmic male-sterile corn anthers
Stamen specification and anther development in rice
Male reproductive development is a complex biological process which includes the formation of the stamen with differentiated anther tissues, in which microspores/pollens are generated, then anther dehiscence and subsequently pollination. Stamen specification and anther development involve a number of extraordinary events such as meristem transition, cell division and differentiation, cell to cell communication, etc., which need the cooperative interaction of sporophytic and gametophytic genes. The advent of various tools for rice functional gene identification, such as complete genome sequence, genome-wide microarrays, collections of mutants, has greatly facilitated our understanding of mechanisms of rice stamen specification and anther development. Male sterile lines are critical for hybrid rice breeding, therefore understanding these processes will not only contribute greatly to the basic knowledge of crop developmental biology, but also to the development of new varieties for hybrid rice breeding in the future.
Cytological analysis and genetic control of rice anther development
icrosporogenesis and male gametogenesis are essential for the alternating life cycle of flowering plants between diploid sporophyte and haploid gametophyte generations.Rice(Oryza saliva) is the world's major staple food,and manipulation of pollen fertility is particularly important for the demands to increase rice grain yield.Towards a better understanding of the mechanisms controlling rice male reproductive development,we describe here the cytological changes of anther development through 14 stages,including cell division,differentiation and degeneration of somatic tissues consisting of four concentric cell layers surrounding and supporting reproductive cells as they form mature pollen grains through meiosis and mitosis.Furthermore,we compare the morphological difference of anthers and pollen grains in both monocot rice and eudicot Arabidopsis thaliana.Additionally,we describe the key genes identified to date critical for rice anther development and pollen formation.
IRREGULAR POLLEN EXINE1 is a novel factor in anther cuticle and pollen exine formation
Anther cuticle and pollen exine are protective barriers for pollen development and fertilization. Despite that several regulators have been identified for anther cuticle and pollen exine development in rice (Oryza sativa) and Arabidopsis (Arabidopsis thaliana), few genes have been characterized in maize (Zea mays) and the underlying regulatory mechanism remains elusive. Here, we report a novel male-sterile mutant in maize, irregular pollen exine1 (ipe1), which exhibited a glossy outer anther surface, abnormal Ubisch bodies, and defective pollen exine. Using map-based cloning, the IPE1 gene was isolated as a putative glucose-methanol-choline oxidoreductase targeted to the endoplasmic reticulum. Transcripts of IPE1 were preferentially accumulated in the tapetum during the tetrad and early uninucleate microspore stage. A biochemical assay indicated that ipe1 anthers had altered constituents of wax and a significant reduction of cutin monomers and fatty acids. RNA sequencing data revealed that genes implicated in wax and flavonoid metabolism, fatty acid synthesis, and elongation were differentially expressed in ipe1 mutant anthers. In addition, the analysis of transfer DNA insertional lines of the orthologous gene in Arabidopsis suggested that IPE1 and their orthologs have a partially conserved function in male organ development. Our results showed that IPE1 participates in the putative oxidative pathway of C16/C18 -hydroxy fatty acids and controls anther cuticle and pollen exine development together with MALE STERILITY26 and MALE STERILITY45 in maize.
ABNORMAL POLLEN VACUOLATION1 (APV1) is required for male fertility by contributing to anther cuticle and pollen exine formation in maize
Abstract Anther cuticle and pollen exine are the major protective barriers against various stresses. The proper functioning of genes expressed in the tapetum is vital for the development of pollen exine and anther cuticle. In this study, we report a tapetum-specific gene, Abnormal Pollen Vacuolation1 (APV1), in maize that affects anther cuticle and pollen exine formation. The apv1 mutant was completely male sterile. Its microspores were swollen, less vacuolated, with a flat and empty anther locule. In the mutant, the anther epidermal surface was smooth, shiny, and plate-shaped compared with the three-dimensional crowded ridges and randomly formed wax crystals on the epidermal surface of the wild type. The wild type mature pollen had elaborate exine patterning, whereas the apv1 pollen surface was smooth. Only a few unevenly distributed Ubisch bodies were formed on the apv1 mutant, leading to a more apparent inner surface. A significant reduction in the cutin monomers was observed in the mutant. APV1 encodes a member of the P450 subfamily, CYP703A2-Zm, which contains 530 amino acids. APV1 appeared to be widely expressed in the tapetum at the vacuolation stage, and its protein signal colocalized with the endoplasmic reticulum (ER) signal. RNA-Seq data revealed that most of the genes in the fatty acid metabolism pathway were differentially expressed in the apv1 mutant. Altogether, we suggest that APV1 functions in the fatty acid hydroxylation pathway which is involved in forming sporopollenin precursors and cutin monomers that are essential for the development of pollen exine and anther cuticle in maize. This article is protected by copyright. All rights reserved.
The transformation of anthers in the msca1 mutant of maize
In normal anther development in maize ( Zea mays L), large hypodermal cells in anther primordia undergo a series of proscribed cell divisions to form an anther containing microsporogenous cells and three distinctive anther wall layers: the tapetum, the middle layer and the endothecium. In homozygous msca1 mutants of maize, stamen primordia are initiated normally and large hypodermal cells can be detected in developing anthers. However, the normal series of cell division and differentiation events does not occur in msca1 mutant plants. Rather, structures containing parenchymal cells and ectopic, nonfunctional vascular strands are formed. The epidermal surfaces of these structures contain stomata, which are normally absent in maize anthers. Thus, all of the cell layers of the "anther" have been transformed in mutant plants. The filaments of the mutant structures are normal, and all other flower parts are normal. The msca1 mutation does not affect female fertility, but transformed "stamen" structures remain associated with mature ovules rather than aborting as in normal ear development. The msca1 mutation is distinctive in that only one part of a single (male) reproductive organ is transformed. The resulting structure has general vegetative features, but cannot be conclusively identified as a particular vegetative organ.
Cloning and Use of the ms9 gene from Maize: US, US20160024520A1. 2016-01-08.[2017-08-19].
MS23, a master basic helix-loop-helix factor, regulates the specification and development of the tapetum in maize
Abstract Successful male gametogenesis involves orchestration of sequential gene regulation for somatic differentiation in pre-meiotic anthers. We report here the cloning of Male Sterile23 (Ms23), encoding an anther-specific predicted basic helix-loop-helix (bHLH) transcription factor required for tapetal differentiation; transcripts localize initially to the precursor secondary parietal cells then predominantly to daughter tapetal cells. In knockout ms23-ref mutant anthers, five instead of the normal four wall layers are observed. Microarray transcript profiling demonstrates a more severe developmental disruption in ms23-ref than in ms32 anthers, which possess a different bHLH defect. RNA-seq and proteomics data together with yeast two-hybrid assays suggest that MS23 along with MS32, bHLH122 and bHLH51 act sequentially as either homo- or heterodimers to choreograph tapetal development. Among them, MS23 is the earliest-acting factor, upstream of bHLH51 and bHLH122, controlling tapetal specification and maturation. By contrast, MS32 is constitutive and independently regulated and is required later than MS23 in tapetal differentiation.
Regulation of cell divisions and differentiation by MALE STERILITY32 is required for anther development in maize
Male fertility in flowering plants relies on proper division and differentiation of cells in the anther, a process that gives rise to four somatic layers surrounding central germinal cells. The maize gene male sterility32 (ms32) encodes a basic helix–loop–helix (bHLH) transcription factor, which functions as an important regulator of both division and differentiation during anther development. After the four somatic cell layers are generated properly through successive periclinal divisions, in the ms32 mutant, tapetal precursor cells fail to differentiate, and, instead, undergo additional periclinal divisions to form extra layers of cells. These cells become vacuolated and expand, and lead to failure in pollen mother cell development. ms32 expression is specific to the pre-meiotic anthers and is distributed initially broadly in the four lobes, but as the anther develops, its expression becomes restricted to the innermost somatic layer, the tapetum. The ms32-ref mac1-1 double mutant is unable to form tapetal precursors and also exhibits excessive somatic proliferation leading to numerous, disorganized cell layers, suggesting a synergistic interaction between ms32 and mac1. Altogether, our results show that MS32 is a major regulator in maize anther development that promotes tapetum differentiation and inhibits periclinal division once a tapetal cell is specified.
Maize Male sterile 8(Ms8), a putative-1,3-galactosyltransferase, modulates cell division, expansion, and differentiation during early maize anther development
Abstract Precise somatic and reproductive cell proliferation and differentiation in anthers are crucial for male fertility. Loss of function of the Male sterile 8 (Ms8) gene causes male sterility with multiple phenotypic defects first visible in the epidermal and tapetal cells. Here, we document the cloning of Ms8, which is a putative -1,3-galactosyltransferase. Ms8 transcript is abundant in immature anthers with a peak at the meiotic stage; RNA expression is highly correlated with protein accumulation. Co-immunoprecipitation coupled with mass spectrometry sequencing identified several MS8-associated proteins, including arabinogalactan proteins, prohibitins, and porin. We discuss the hypotheses that arabinogalactan protein might be an MS8 substrate and that MS8 might be involved in maintenance of mitochondrial integrity.
Nucleotide Sequences Mediated Male Fertility and Method of Using Same: United States, US2012/0005792 A1
2012-01-05. [2017-08-19].[2017-08-19]..
Nucleotide Sequences Mediated Male Fertility and Method of Using Same: United States, US005478369A.1995-01-13.[2017-08-19].
The Maize Male Gametophyte//Bennetzen J L, Hake. Handbook of Maize: Its Biology
Specification of tapetum and microsporocyte cells within the anther
Flowering plants form male reproductive cells (microsporocytes) during sporophytic generation, which subsequently differentiate into multicellular male gametes in the gametophytic generation. The tapetum is a somatic helper tissue neighboring microsporocytes and supporting gametogenesis. The mechanism controlling the specification of the tapetum and microsporocyte cell fate within the anther has long been a mystery in biology. Recent investigations have revealed molecular switches and signaling pathways underlying the establishment of somatic and reproductive cells in plants. In this review we discuss common and diversified signaling molecules and regulatory pathways including receptor-like protein kinases, redox status, glycoprotein, transcription factors, hormones and microRNA implicated in the specification of tapetum and microsporocytes in plants.
The mac1 mutation alters the developmental fate of the hypodermal cells and their cellular progeny in the maize anther
Abstract In angiosperm ovules and anthers, the hypodermal cell layer provides the progenitors of meiocytes. We have previously reported that the multiple archesporial cells1 (mac1) mutation identifies a gene that plays an important role in the switch of the hypodermal cells from the vegetative pathway to the meiotic (sporogenous) pathway in maize ovules. Here we report that the mac1 mutation alters the developmental fate of the hypodermal cells of the maize anther. In a normal anther a hypodermal cell divides periclinally with the inner cell giving rise to the sporogenous archesporial cells while the outer cell, together with adjacent cells, forms the primary parietal layer. The cells of the parietal layer then undergo two cycles of periclinal divisions to give rise to three wall layers. In mac1 anthers the primary parietal layer usually fails to divide periclinally so that the three wall layers do not form, while the archesporial cells divide excessively and most fail to form microsporocytes. The centrally located mutant microsporocytes are abnormal in appearance and in callose distribution and they fail to proceed through meiosis. These failures in development and function appear to reflect the failure of mac1 gene function in the hypodermal cells and their cellular progeny.
Cytological characterization and allelism testing of anther developmental mutants identified in a screen of maize male sterile lines
Proper regulation of anther differentiation is crucial for producing functional pollen, and defects in or absence of any anther cell type result in male sterility. To deepen understanding of processes required to establish premeiotic cell fate and differentiation of somatic support cell layers a cytological screen of maize male-sterile mutants has been conducted which yielded 42 new mutants including 22 mutants with premeiotic cytological defects (increasing this class fivefold), 7 mutants with postmeiotic defects, and 13 mutants with irregular meiosis. Allelism tests with known and new mutants confirmed new alleles of four premeiotic developmental mutants, including two novel alleles of msca1 and single new alleles of ms32, ms8, and ocl4, and two alleles of the postmeiotic ms45. An allelic pair of newly described mutants was found. Premeiotic mutants are now classified into four categories: anther identity defects, abnormal anther structure, locular wall defects and premature degradation of cell layers, and/or microsporocyte collapse. The range of mutant phenotypic classes is discussed in comparison with developmental genetic investigation of anther development in rice and Arabidopsis to highlight similarities and differences between grasses and eudicots and within the grasses.
Molecular and genetic analyses of the Silky1 gene reveal conservation in floral organ specification between eudicots and monocots
The degree to which the eudicot-based ABC model of flower organ identity applies to the other major subclass of angrosperms, the monocots, has yet to be fully explored. We cloned silky1 (si1), a male sterile mutant of Zea mays that has homeotic conversions of stamens into carpels and lodicules into palea/lemma-like structures. Our studies indicate that si1 is a monocot B function MADS box gene. Moreover, the si1 zag1 double mutant produces a striking spikelet phenotype where normal glumes enclose reiterated palea/lemma-like organs. These studies indicate that B function gene activity is conserved among monocots as well as eudicots. In addition, they provide compelling developmental evidence for recognizing lodicules as modified petals and, possibly, palea and lemma as modified sepals.
The maize PI/GLO ortholog Zmm16/sterile tassel silky ear1 interacts with the zygomorphy and sex determination pathways in flower development
.
Two male-sterile mutants of Zea mays with an extra cell division in anther wall
Two recessive male-sterile mutants of maize with similar patterns of pollen abortion were studied. Genetic studies showed that one of the two mutations was allelic with a previously identified male-sterility locus (ms23) and the other mutation was in a newly identified male-sterility locus (ms32). Cytological characterization of homozygous mutants and fertile heterozygous control siblings was performed using brightfield, fluorescence, and electron microscopy. During normal anther development, the final anther wall periclinal division divides the secondary parietal anther wall layer into the middle layer and tapetum, forming an anther with four wall layers. This is followed by differentiation of the tapetal cells into protoplastic binucleate, secretory tissue. In both the ms23 and ms32 mutants, the prospective tapetal layer divided into two layers, termed t1 and t2, forming an anther with five wall layers. Neither the t1 nor the t2 layers differentiated normally into tapetal layers, as determined by examination of cell walls, nucleus number, and cytoplasmic organization. Pollen mother cells aborted after the onset of prophase I of meiosis, suggesting that an early developmental coordination may exist between tapetum and pollen mother cells.
Biosynthesis and secretion of plant cuticular wax
Abstract The cuticle covers the aerial portions of land plants. It consists of amorphous intracuticular wax embedded in cutin polymer, and epicuticular wax crystalloids that coat the outer plant surface and impart a whitish appearance. Cuticular wax is mainly composed of long-chain aliphatic compounds derived from very long chain fatty acids. Wax biosynthesis begins with fatty acid synthesis in the plastid. Here we focus on fatty acid elongation (FAE) to very long chains (C24-C34), and the subsequent processing of these elongated products into alkanes, secondary alcohols, ketones, primary alcohols and wax esters. The identity of the gene products involved in these processes is starting to emerge. Other areas of this field remain enigmatic. For example, it is not known how the hydrophobic wax components are moved intracellularly, how they are exported out of the cell, or translocated through the hydrophilic cell wall. Two hypotheses are presented for intracellular wax transport: direct transfer of lipids from the endoplasmic reticulum to the plasma membrane, and Golgi mediated exocytosis. The potential roles of ABC transporters and non-specific lipid transfer proteins in wax export are also discussed. Biochemical-genetic and genomic approaches in Arabidopsis thaliana promise to be particularly useful in identifying and characterizing gene products involved in wax biosynthesis, secretion and function. The current review will, therefore, focus on Arabidopsis as a model for studying these processes.
The formation and function of plant cuticles
The plant cuticle is an extracellular hydrophobic layer that covers the aerial epidermis of all land plants, providing protection against desiccation and external environmental stresses. The past decade has seen considerable progress in assembling models for the biosynthesis of its two major components, the polymer cutin and cuticular waxes. Most recently, two breakthroughs in the long-sought molecular bases of alkane formation and polyester synthesis have allowed construction of nearly complete biosynthetic pathways for both waxes and cutin. Concurrently, a complex regulatory network controlling the synthesis of the cuticle is emerging. It has also become clear that the physiological role of the cuticle extends well beyond its primary function as a transpiration barrier, playing important roles in processes ranging from development to interaction with microbes. Here, we review recent progress in the biochemistry and molecular biology of cuticle synthesis and function and highlight some of the major questions that will drive future research in this field.
Sporopollenin monomer biosynthesis in arabidopsis
Land plants have evolved aliphatic biopolymers that protect their cell surfaces against dehydration, pathogens, and chemical and physical damage. In flowering plants, a critical event during pollen maturation is the formation of the pollen surface structure. The pollen wall consists essentially of the microspore-derived intine and the sporophyte-derived exine. The major component of the exine is termed sporopollenin, a complex biopolymer. The chemical composition of sporopollenin remains poorlycharacterized because it is extremely resistant to chemical and biological degradation procedures. Recent characterization of Arabidopsis thaliana genes and corresponding enzymes involved in exine formation has demonstrated that the sporopollenin polymer consists of phenolic and fatty acid-derived constituents that are covalently coupled by ether and ester linkages. This review illuminates the outlines of a biosynthetic pathway involved in generating monomer constituents of the sporopollenin biopolymer component of the pollen wall.
Cytochrome P450 family member CYP704B2 catalyzes the -hydroxylation of fatty acids and is required for anther cutin biosynthesis and pollen exine formation in rice
Defective pollen wall is required for anther and microspore development in rice and encodes a fatty acyl carrier protein reductase
Aliphatic alcohols naturally exist in many organisms as important cellular components; however, their roles in extracellular polymer biosynthesis are poorly defined. We report here the isolation and characterization of a rice (Oryza sativa) male-sterile mutant, defective pollen wall (dpw), which displays defective anther development and degenerated pollen grains with an irregular exine. Chemical analysis revealed that dpw anthers had a dramatic reduction in cutin monomers and an altered composition of cuticular wax, as well as soluble fatty acids and alcohols. Using map-based cloning, we identified the DPW gene, which is expressed in both tapetal cells and microspores during anther development. Biochemical analysis of the recombinant DPW enzyme shows that it is a novel fatty acid reductase that produces 1-hexadecanol and exhibits >270-fold higher specificity for palmiltoyl-acyl carrier protein than for C16:0 CoA substrates. DPW was predominantly targeted to plastids mediated by its N-terminal transit peptide. Moreover, we demonstrate that the monocot DPW from rice complements the dicot Arabidopsis thaliana male sterile2 (ms2) mutant and is the probable ortholog of MS2. These data suggest that DPWs participate in a conserved step in primary fatty alcohol synthesis for anther cuticle and pollen sporopollenin biosynthesis in monocots and dicots.
Comparative profiling of the sense and antisense transcriptome of maize lines
There are thousands of maize lines with distinctive normal as well as mutant phenotypes. To determine the validity of comparisons among mutants in different lines, we first address the question of how
Differential gene expression in Arabidopsis wild-type and mutant anthers: insights into anther cell differentiation and regulatory networks
In flowering plants, the anther contains highly specialized reproductive and somatic cells that are required for male fertility. Genetic studies have uncovered several genes that are important for anther development. However, little information is available regarding most genes active during anther development, including possible relationships between these genes and genetically defined regulators. In Arabidopsis, two previously isolated male-sterile mutants display dramatically altered anther cell differentiation patterns. The sporocyteless (spl)/nozzle (nzz) mutant is defective in the differentiation of primary sporogenous cells into microsporocytes, and does not properly form the anther wall. The excess microsporocytes1 (ems1)/extrasporogenous cells (exs) mutants produce excess microsporocytes at the expense of the tapetum. To gain additional insights into microsporocyte and tapetum differentiation and to uncover potential genetic interactions, expression profiles were compared between wild-type anthers (stage 4-6) and those of the spl or ems1 mutants. A total of 1954 genes were found to be differentially expressed in the ems1 and/or spl anthers, and these were grouped into 14 co-expression clusters. The presence of genes with known and predicted functions in specific clusters suggests potential functions for other genes in the same cluster. To obtain clues about possible co-regulation within co-expression clusters, we searched for shared cis-regulatory motifs in putative promoter regions. Our analyses were combined with data from previous studies to develop a model of the anther gene regulatory network. This model includes hypotheses that can be tested experimentally to gain further understanding of the mechanisms controlling anther development.
Mutator transposon activity reprograms the transcriptomes and proteomes of developing maize anthers
Summary Despite the high conservation of anther gene expression patterns across maize lines, Mu transposition programmed by transcriptionally active MuDR results in a 25% change in the transcriptome, monitored over 90h of immature anther development, without altering the morphology, anatomy or pace of development. Most transcriptome changes are stage specific: cases of suppression of normal transcripts and ectopic activation are equally represented. Protein abundance changes were validated for numerous metabolic enzymes, and highlight the increased carbon and reactive oxygen management in Mutator anthers. Active Mutator lines appear to experience chronic stress, on a par with abiotic treatments that stimulate early flowering. Despite the diversity of acclimation responses, anther development progresses normally, in contrast to male-sterile mutants that disrupt anther cell fate or function completely, and cause fewer transcriptome changes. The early flowering phenotype ultimately confers an advantage in Mu element transmission.
Transcriptome profiling of maize anthers using genetic ablation to analyze pre-meiotic and tapetal cell types
.Oligonucleotide arrays were used to profile gene expression in dissected maize anthers at four stages: after-anther initiation, at the rapid mitotic proliferation stage, pre-meiosis, and meiotic prophase I. Nearly 9200 sense and antisense transcripts were detected, with the most diverse transcriptome present at the pre-meiotic stage. Three male-sterile mutants lacking a range of normal cell types resulting from a temporal progression of anther failure were compared with fertile siblings at equivalent stages by transcription profiles. The msca1 mutant has the earliest visible phenotype, develops none of the normal anther cell types and exhibits the largest deviation from fertile siblings. The mac1 mutant has an excess of archesporial derivative cells and lacks a tapetum and middle layer, resulting in moderate transcriptional deviations. The ms23 mutant lacks a differentiated tapetum and shows the fewest differences from fertile anthers. By combining the data sets from the comparisons between individual sterile and fertile anthers, candidate genes predicted to play important roles during maize anther development were assigned to stages and to likely cell types. Comparative analyses with a data set of anther-specific genes from rice highlight remarkable quantitative similarities in gene expression between these two grasses.
Transcriptomes and proteomes define gene expression progression in pre-meiotic maize anthers
Plants lack a germ line; consequently, during reproduction adult somatic cells within flowers must switch from mitotic proliferation to meiosis. In maize (Zea mays L.) anthers, hypoxic conditions in the developing tassel trigger pre-meiotic competence in the column of pluripotent progenitor cells in the center of anther lobes, and within 24 hr these newly specified germinal cells have patterned their surrounding neighbors to differentiate as the first somatic niche cells. Transcriptomes were analyzed by microarray hybridization in carefully staged whole anthers during initial specification events, after the separation of germinal and somatic lineages, during the subsequent rapid mitotic proliferation phase, and during final pre-meiotic germinal and somatic cell differentiation. Maize anthers exhibit a highly complex transcriptome constituting nearly three-quarters of annotated maize genes, and expression patterns are dynamic. Laser microdissection was applied to begin assigning transcripts to tissue and cell types and for comparison to transcriptomes of mutants defective in cell fate specification. Whole anther proteomes were analyzed at three developmental stages by mass spectrometric peptide sequencing using size-fractionated proteins to evaluate the timing of protein accumulation relative to transcript abundance. New insights include early and sustained expression of meiosis-associated genes (77.5% of well-annotated meiosis genes are constitutively active in 0.15 mm anthers), an extremely large change in transcript abundances and types a few days before meiosis (including a class of 1340 transcripts absent specifically at 0.4 mm), and the relative disparity between transcript abundance and protein abundance at any one developmental stage (based on 1303 protein-to-transcript comparisons).
Metabolic profiling: pathways in discovery
Highlights of Cambridge Healthtech Institute's 3rd annual Metabolic Profiling: Pathways in Discovery conference, held on 8-9 December 2003 at the Hyatt Regency Princeton, NJ, USA.
The secondary metabolism of Arabidopsis thaliana: growing like a weed
Genetics and biochemistry of secondary metabolites: an evolutionary perspective
The evolution of new genes to make novel secondary compounds in plants is an ongoing process and might account for most of the differences in gene function among plant genomes. Although there are many substrates and products in plant secondary metabolism, there are only a few types of reactions. Repeated evolution is a special form of convergent evolution in which new enzymes with the same function evolve independently in separate plant lineages from a shared pool of related enzymes with similar but not identical functions. This appears to be common in secondary metabolism and might confound the assignment of gene function based on sequence information alone.
Arabidopsis cuticular waxes: advances in synthesis, export and regulation
Cuticular waxes and cutin form the cuticle, a hydrophobic layer covering the aerial surfaces of land plants and acting as a protective barrier against environmental stresses. Very-long-chain fatty acid derived compounds that compose the cuticular waxes are produced in the endoplasmic reticulum of epidermal cells before being exported to the environmental face of the epidermis. Twenty years of genetic studies on Arabidopsis thaliana have led to the molecular characterization of enzymes catalyzing major steps in fatty acid elongation and wax biosynthesis. Although transporters required for wax export from the plasma membrane have been identified, intracellular and extracellular traffic remains largely unknown. In accordance with its major function in producing an active waterproof barrier, wax metabolism is up-regulated at the transcriptional level in response to water deficiency. However its developmental regulation is still poorly described. Here, we discuss the present knowledge of wax functions, biosynthesis and transport as well as the regulation of these processes.
Biophysical and biochemical characteristics of cutin, a plant barrier biopolymer
Abstract Cutin is a support biopolyester involved in waterproofing the leaves and fruits of higher plants, regulating the flow of nutrients among various plant cells and organs, and minimizing the deleterious impact of pathogens. Despite the complexity and intractable nature of this biopolymer, significant progress in chemical composition, molecular architecture and, more recently, biosynthesis have been made in the past 10 years. This review is focused in the description of these advances and their physiological impacts to improve our knowledge on plant cutin, an unusual topic in most plant physiology and biochemistry books and reviews.
Sporopollenin monomer biosynthesis in arabidopsis
Land plants have evolved aliphatic biopolymers that protect their cell surfaces against dehydration, pathogens, and chemical and physical damage. In flowering plants, a critical event during pollen maturation is the formation of the pollen surface structure. The pollen wall consists essentially of the microspore-derived intine and the sporophyte-derived exine. The major component of the exine is termed sporopollenin, a complex biopolymer. The chemical composition of sporopollenin remains poorlycharacterized because it is extremely resistant to chemical and biological degradation procedures. Recent characterization of Arabidopsis thaliana genes and corresponding enzymes involved in exine formation has demonstrated that the sporopollenin polymer consists of phenolic and fatty acid-derived constituents that are covalently coupled by ether and ester linkages. This review illuminates the outlines of a biosynthetic pathway involved in generating monomer constituents of the sporopollenin biopolymer component of the pollen wall.
Molecular phenotyping of the pal1 and pal2 mutants of Arabidopsis thaliana reveals far-reaching consequences on phenylpropanoid, amino acid, and carbohydrate metabolism
.
Spatiotemporally dynamic, cell-type-dependent premeiotic and meiotic phasiRNAs in maize anthers
PMS1T, producing phased small-interfering RNAs, regulates photoperiod-sensitive male sterility in rice
Phased small-interfering RNAs (phasiRNAs) are a special class of small RNAs, which are generated in 21- or 24-nt intervals from transcripts of precursor RNAs. Although phasiRNAs have been found in a range of organisms, their biological functions in plants have yet to be uncovered. Here we show that phasiRNAs generated by the photopheriod-sensetive genic male sterility 1 (Pms1) locus were associated with photoperiod-sensitive male sterility (PSMS) in rice, a germplasm that started the two-line hybrid rice breeding. The Pms1 locus encodes a long-noncoding RNA PMS1T that was preferentially expressed in young panicles. PMS1T was targeted by miR2118 to produce 21-nt phasiRNAs that preferentially accumulated in the PSMS line under long-day conditions. A single nucleotide polymorphism in PMS1T nearby the miR2118 recognition site was critical for fertility change, likely leading to differential accumulation of the phasiRNAs. This result suggested possible roles of phasiRNAs in reproductive development of rice, demonstrating the potential importance of this RNA class as regulators in biological processes.
Identification of MIR390a precursor processing-defective mutants in Arabidopsis by directgenome sequencing
Transacting siRNA (tasiRNA) biogenesis in Arabidopsis is initiated by microRNA (miRNA) -guided cleavage of primary transcripts. In the case of TAS3 tasiRNA formation, ARGONAUTE7 (AGO7)-miR390 complexes interact with primary transcripts at two sites, resulting in recruitment of RNA-DEPENDENT RNA POLYMERASE6 for dsRNA biosynthesis. An extensive screen for Arabidopsis mutants with specific defects in TAS3 tasiRNA biogenesis or function was done. This yielded numerous ago7 mutants, one dcl4 mutant, and two mutants that accumulated low levels of miR390. A direct genome sequencing-based approach to both map and rapidly identify one of the latter mutant al铆eles was developed. This revealed a G-to-A point mutation (mir390a-1) that was calculated to stabilize a relatively nonpaired region near the base of the MIR390a foldback, resulting in misprocessing of the miR390/miR390* duplex and subsequent reduced TAS3 tasiRNA levels. Directed substitutions, as well as analysis of variation at paralogous miR390-generating loci (MIR390a and MIR390b), indicated that base pair properties and nucleotide identity within a region 4-6 bases below the miR390/miR390* duplex region contributed to the efficiency and accuracy of precursor processing.
Genetic regulation of sporopollenin synthesis and pollen exine development. Annual Review of
Pollen acts as a biological protector of male sperm and is covered by an outer cell wall polymer called the exine, which consists of durable sporopollenin. Despite the astonishingly divergent structure of the exine across taxa, the developmental processes of its formation surprisingly do not vary, which suggests the preservation of a common molecular mechanism. The precise molecular mechanisms underlying pollen exine patterning remain highly elusive, but they appear to be dependent on at least three major developmental processes: primexine formation, callose wall formation, and sporopollenin synthesis. Several lines of evidence suggest that the sporopollenin is built up via catalytic enzyme reactions in the tapetum, and both the primexine and callose wall provide an efficient substructure for sporopollenin deposition. Herein, we review the currently accepted understanding of the molecular regulation of sporopollenin biosynthesis and examine unanswered questions regarding the requirements underpinning proper exine pattern formation, as based on genetic evidence.
The biosynthesis, composition and assembly of the outer pollen wall: A tough case to crack
The formation of the durable outer pollen wall, largely composed of sporopollenin, is essential for the protection of the male gametophyte and plant reproduction. Despite its apparent strict conservation amongst land plants, the composition of sporopollenin and the biosynthetic pathway(s) yielding this recalcitrant biopolymer remain elusive. Recent molecular genetic studies in Arabidopsis thaliana (Arabidopsis) and rice have, however, identified key genes involved in sporopollenin formation, allowing a better understanding of the biochemistry and cell biology underlying sporopollenin biosynthesis and pollen wall development. Herein, current knowledge of the biochemical composition of the outer pollen wall is reviewed, with an emphasis on enzymes with characterized biochemical activities in sporopollenin and pollen coat biosynthesis. The tapetum, which forms the innermost sporophytic cell layer of the anther and envelops developing pollen, plays an essential role in sporopollenin and pollen coat formation. Recent studies show that several tapetum-expressed genes encode enzymes that metabolize fatty acid derived compounds to form putative sporopollenin precursors, including tetraketides derived from fatty acyl-CoA starter molecules, but analysis of mutants defective in pollen wall development indicate that other components are also incorporated into sporopollenin. Also highlighted are the many uncertainties remaining in the development of a sporopollenin-fortified pollen wall, particularly in relation to the mechanisms of sporopollenin precursor transport and assembly into the patterned form of the pollen wall. A working model for sporopollenin biosynthesis is proposed based on the data obtained largely from studies of Arabidopsis, and future challenges to complete our understanding of pollen wall biology are outlined.
Genetic and biochemical mechanisms of pollen wall development
Abstract The pollen wall is a specialized extracellular cell wall matrix that surrounds male gametophytes and plays an essential role in plant reproduction. Uncovering the mechanisms that control the synthesis and polymerization of the precursors of pollen wall components has been a major research focus in plant biology. We review current knowledge on the genetic and biochemical mechanisms underlying pollen wall development in eudicot model Arabidopsis thaliana and monocot model rice (Oryza sativa), focusing on the genes involved in the biosynthesis, transport, and assembly of various precursors of pollen wall components. The conserved and divergent aspects of the genes involved as well as their regulation are addressed. Current challenges and future perspectives are also highlighted.
水稻雄性发育功能基因的发掘及应用
Functional research of rice male reproduction and its utilization in breeding
Targeted mutagenesis in ea mays using TALENs and the CRISPR/Cas system
Transcription activator-like effector nucleases (TALENs) and clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated (Cas) systems have emerged as powerful tools for genome editing in a variety of species. Here, we report, for the first time, targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas system. We designed five TALENs targeting 4 genes, namely ZmPDS, ZmIPK1A, ZmIPK, ZmMRP4, and obtained targeting efficiencies of up to 23.1% in protoplasts, and about 13.3% to 39.1% of the transgenic plants were somatic mutations. Also, we constructed two gRNAs targeting the ZmIPK gene in maize protoplasts, at frequencies of 16.4% and 19.1%, respectively. In addition, the CRISPR/Cas system induced targeted mutations in Z. mays protoplasts with efficiencies (13.1%) similar to those obtained with TALENs (9.1%). Our results show that both TALENs and the CRISPR/Cas system can be used for genome modification in maize.
Characterization of maize male sterile 2 mutant by phenotypic and RNA sequencing analyses
Abstract Male sterility plays an important role in the hybrid seed production. Elucidation of the mechanism of male sterility will provide useful information for the usage of male sterility. In this study, we performed detailed phenotypic analysis of ms2 mutant, and the results showed that ms2 anthers are defects in synthesis and transportation of lipophilic molecules. To investigate the defects of transcriptome of ms2 anthers, 1.5, 2.5 and 3.5 mm anthers were sampled, respectively. Among the 23 037 expressed transcripts at 1.5 mm stage, only 15 genes were differentially expressed between fertile siblings and ms2 mutant. Compared to the fewer detected differentially expressed genes (DEGs) at 1.5 mm stage, 950 DEGs and 2212 DEGs between the fertile siblings and ms2 anthers were detected at 2.5 mm and 3.5 mm stages. These differentially expressed genes (DEGs) were mainly enriched in the secondary metabolism and lipid metabolism pathways. A series of genes related to the long-chain fatty acids metabolism process were changed in ms2 mutant compared with the fertile siblings. The results in this study provide useful information on the mechanism of male sterility in ms2 mutant.
Construction of a multi-control sterility system for a maize male-sterile line and hybrid seed production based on the ZmMs7 gene encoding a PHD-finger transcription factor
Although hundreds of genetic male sterility (GMS) mutants have been identified in maize, few are commercially used due to a lack of effective methods to produce large quantities of pure male‐sterile seeds. Here, we develop a multi‐control sterility (MCS) system based on the maize male sterility 7 (ms7) mutant and its wild‐type Zea mays Male sterility 7 (ZmMs7) gene via a transgenic strategy, leading to the utilization of GMS in hybrid seed production. ZmMs7 is isolated by a map‐based cloning approach, and encodes a PHD‐finger transcription factor orthologous to rice PTC1 and Arabidopsis MS1. The MCS transgenic maintainer lines are developed based on the ms7‐6007 mutant transformed with MCS constructs containing the (i) ZmMs7 gene to restore fertility, (ii) α‐amylase gene ZmAA and/or (iii) DNA adenine methylase gene Dam to devitalize transgenic pollen, (iv) red fluorescence protein gene DsRed2 or mCherry to mark transgenic seeds, and (v) herbicide‐resistant gene Bar for transgenic seed selection. Self‐pollination of the MCS transgenic maintainer line produces transgenic red fluorescent seeds and non‐transgenic normal color seeds at a 1:1 ratio. Among them, all the fluorescent seeds are male fertile, but the seeds with a normal color are male sterile. Cross‐pollination of the transgenic plants to male‐sterile plants propagates male‐sterile seeds with high purity. Moreover, the transgene transmission rate through pollen of transgenic plants harboring two pollen‐disrupted genes is lower than that containing one pollen‐disrupted gene. The MCS system has great potential to enhance the efficiency of maize male‐sterile line propagation and commercial hybrid seed production.